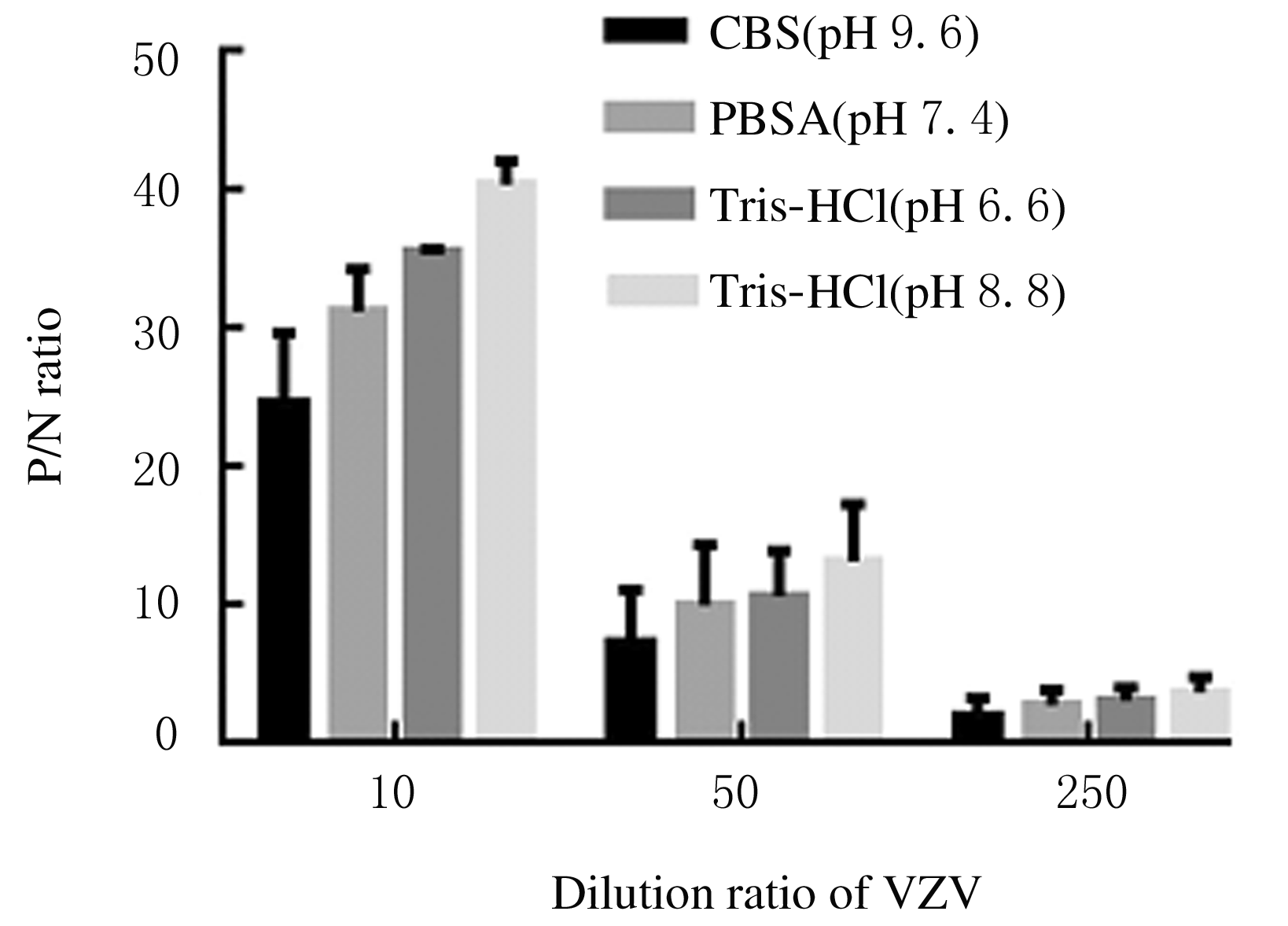
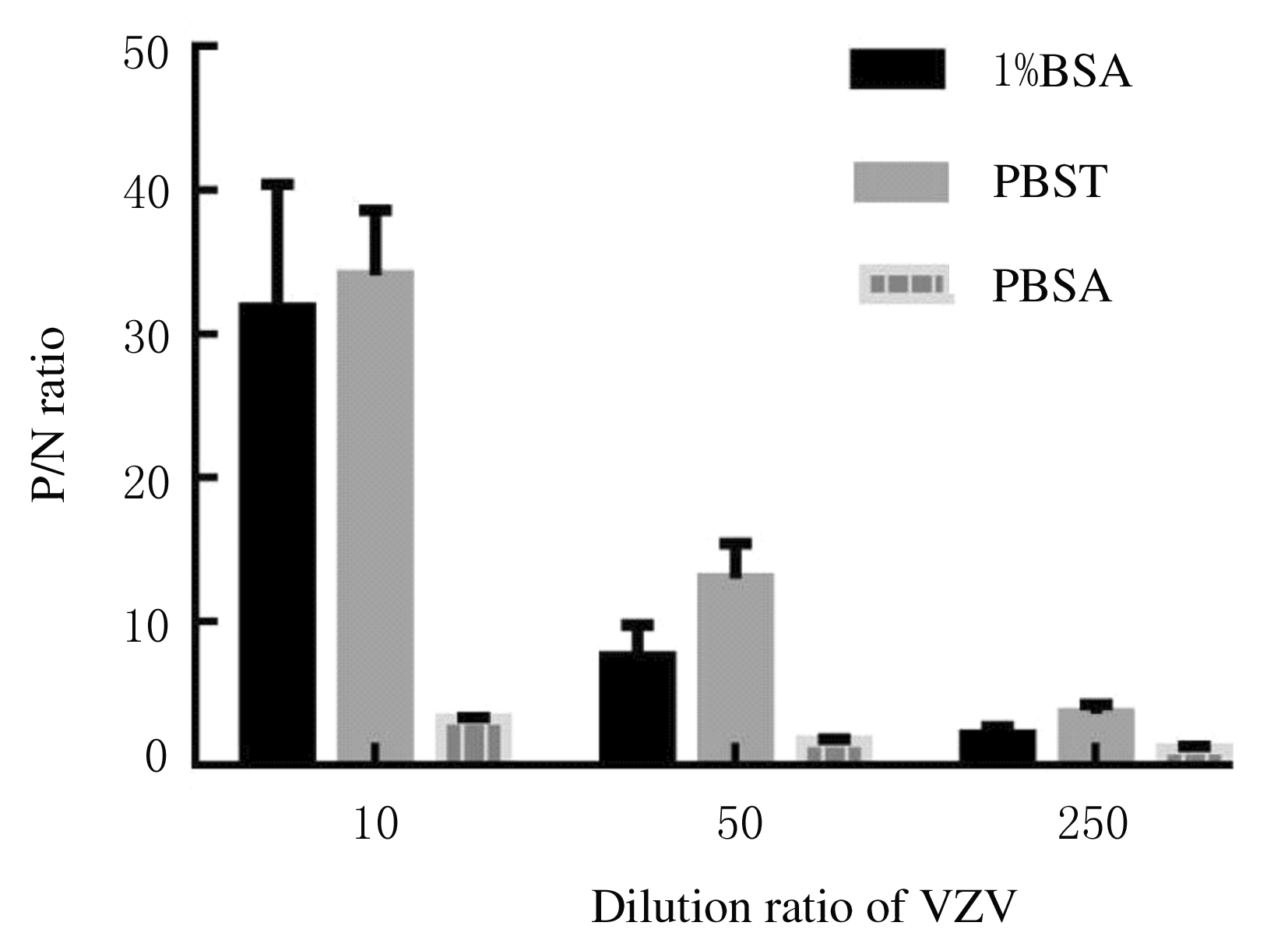
| [1] |
Mengyao SHAN,Weigang WANG,Jinxiang DONG,Jianming TIAN,Lianlian SONG,Yinghong CHEN,Xiaoxue FANG,Zhidong QIU,Haoming LUO,Difu ZHU.
Improvement effect of Panax ginseng glycoproteins on spermatogenesis of mice with oligoasthenozoospermia
[J]. Journal of Jilin University(Medicine Edition), 2022, 48(3): 575-583.
|
| [2] |
Yinong LIU,Qiang ZHANG,Li XU.
Improvement effect of atorvastatin on vascular endothelial dysfunction induced by Ox-LDL/β2GPⅠ/anti-β2GPⅠ complex and its mechanism
[J]. Journal of Jilin University(Medicine Edition), 2022, 48(2): 317-323.
|
| [3] |
Qian MAO, Cuicui CHEN, Huankun LIANG, Penge LIU, Shuhai ZHONG, Laiqing LI.
Establishment and evaluation of a double?labeled time? resolved immunofluorescence analysis method for detecting levels of β?CTX and N?MID
[J]. Journal of Jilin University(Medicine Edition), 2021, 47(3): 770-776.
|
| [4] |
LIU Wei, LUO Xiaohua, CHEN Wenjing, DONG Yuan, ZHU Jie, GUO Jian, JIANG Yong, ZHANG Lei, MENG Guixian, WANG Huiyan.
Expression and immunization assessment of HSV-2 glycoprotein D in baculovirus expression vector system
[J]. Journal of Jilin University Medicine Edition, 2017, 43(04): 720-724.
|
| [5] |
LUO Haoming, CHEN Yinghong, ZHOU Tingting, HONG Tie, JIANG Ruizhi, WANG Ying, YANG Xiaohong, MA Li.
Influence of glycoprotein from Panax ginsengin study and memory abilities in mice
[J]. Journal of Jilin University Medicine Edition, 2016, 42(03): 439-445.
|
| [6] |
JIANG Hui, ZHANG Tongfei, YANG He, XU Zhaonan, BI Ye, SUN Shu, ZHANG Zebing, JIA Jie.
Establishment of human tongue squamous cell carcinoma cisplatin resistant cell line CAL-27/DDPand its biological evaluation
[J]. Journal of Jilin University Medicine Edition, 2016, 42(03): 506-511.
|
| [7] |
LI Jing,LIANG Xiao-fang,ZHAI Gui-lan .
Application of combined detection of serum levels of CA199,
CA125 and CEA in diagnosis of pancreatic cancer
[J]. Journal of Jilin University Medicine Edition, 2014, 40(06): 1252-.
|
| [8] |
SUN Qin-nuan,LI Dong-mei,YU Hui-ling,MA Hong-xi.
Expressions of inflammatory factors in thyroid papillary
carcinoma tissue and their clinical significances
[J]. Journal of Jilin University Medicine Edition, 2014, 40(06): 1261-1265.
|
| [9] |
ZHAO Hai-Yan, ZHANG Xiao-Li, GUO Yun, JIANG Li, XIAO Wei-Guo.
Determination of a proliferation-inducing ligand |in patients with systemic lupus erythematosus and its clinical significance
[J]. J4, 2011, 37(1): 105-108.
|
| [10] |
ZENG Fei-Yue, WANG Yan-Jin, CHEN Feng-Hua, YANG Zhuan-Yi, DENG Yong-Wen, FANG Jia-Sheng.
Expression and significance of MDR1 in human brain |tumor stem cells
[J]. J4, 2009, 35(5): 769-773.
|
| [11] |
FU Jia, TAN Yan, FANG Yan-Qiu, XIONG Bin, XU Li.
Preliminary approach on application of recombinant human |2-glycoprotein-1 domain Ⅰ dimer as B lymphocyte tolerogen
[J]. J4, 2009, 35(4): 646-649.
|
| [12] |
YU Fang,HE Bing,QIU Jun,MA Fang-qin, ZHANG Hong.
Initial evaluation on safety of herpes simplex virus Ⅰglycoprotein B DNA vaccine in mice
[J]. J4, 2008, 34(5): 806-809.
|
| [13] |
PAN Yu-zhuo,ZHAO Yan-ying,ZHAO Xue-jian,LI Yang.
Establishment of multidrug resistance cell line from human prostatic cancinoma
[J]. J4, 2007, 33(4): 651-654.
|
| [14] |
LI Guang-yuan,HE Bing,FENG Fei,MENG Xiang-jun,SONG Yi-shu,WANG Xiao-qi.
Construction and cellular immune response of herpes simplex virus type 1 truncated glycoprotein B DNA vaccine in mice
[J]. J4, 2007, 33(3): 454-459.
|
| [15] |
LIU Chang-jian, LIU Jian-guo, YU Tie-cheng, LUO Zong-jian.
Reversal effect of Tan microemulsion on multidrug resistance
[J]. J4, 2007, 33(2): 293-296.
|
 ),Chunlai JIANG4(
),Chunlai JIANG4( )
)