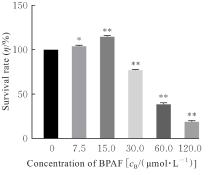
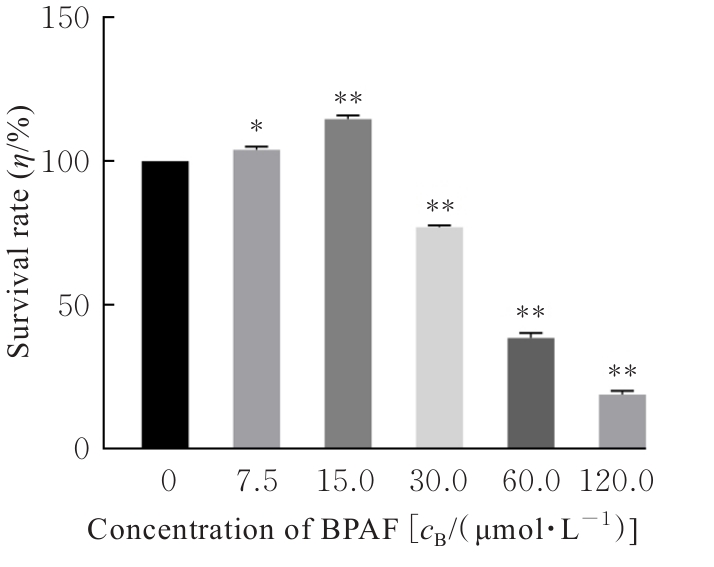
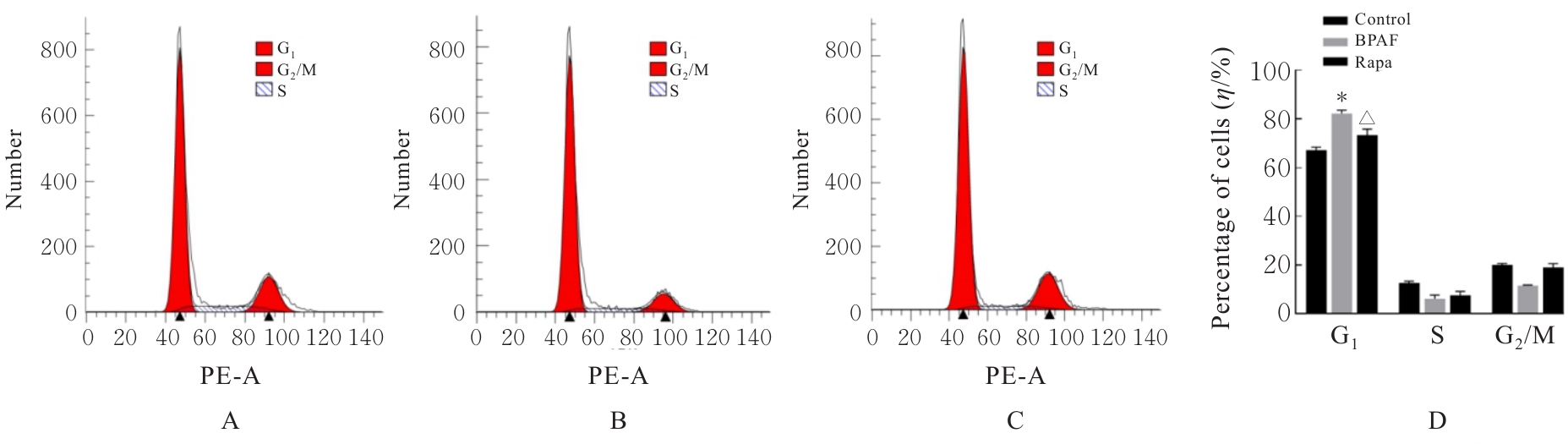
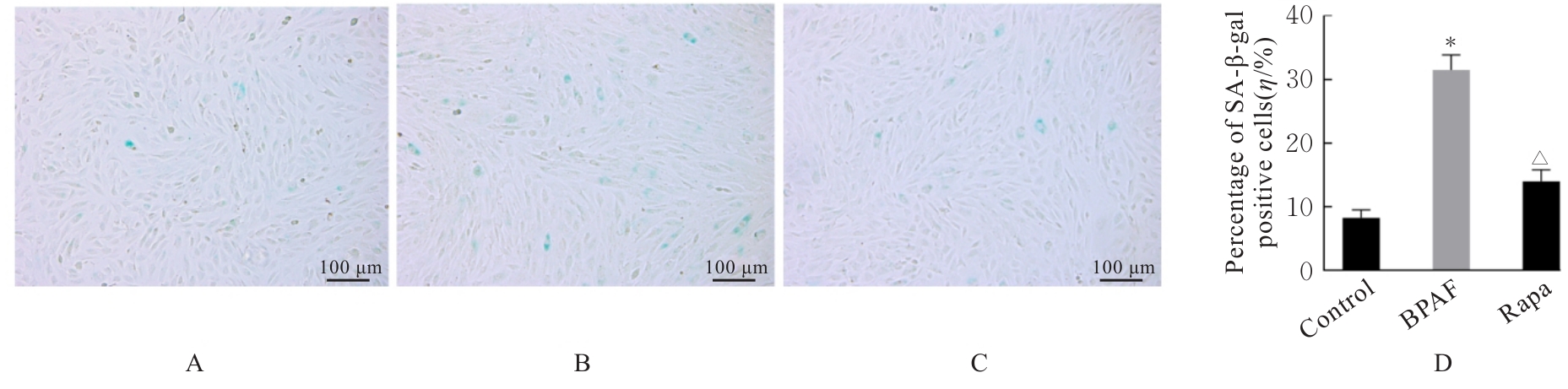
| [1] |
GRIUKOVA A, DERYABIN P, SHATROVA A, et al. Molecular basis of senescence transmitting in the population of human endometrial stromal cells[J]. Aging, 2019, 11(21): 9912-9931.
|
| [2] |
MARUYAMA T, YOSHIMURA Y. Molecular and cellular mechanisms for differentiation and regeneration of the uterine endometrium[J]. Endocr J, 2008, 55(5): 795-810.
|
| [3] |
DERYABIN P I, BORODKINA A V. Stromal cell senescence contributes to impaired endometrial decidualization and defective interaction with trophoblast cells[J]. Hum Reprod, 2022, 37(7): 1505-1524.
|
| [4] |
GORE A C, CHAPPELL V A, FENTON S E, et al. EDC-2: the endocrine society’s second scientific statement on endocrine-disrupting chemicals[J]. Endocr Rev, 2015, 36(6): E1-E150.
|
| [5] |
WEN X, XIONG Y, GENG T, et al. BPA modulates the WDR5/TET2 complex to regulate ERβ expression in eutopic endometrium and drives the development of endometriosis[J]. Environ Pollut, 2021, 268(Pt B): 115748.
|
| [6] |
OLSON M R, SU R W, FLAWS J A, et al. Bisphenol A impairs decidualization of human uterine stromal fibroblasts[J]. Reprod Toxicol, 2017, 73: 339-344.
|
| [7] |
HUANG M Q, LIU S, FU L, et al. Bisphenol A and its analogues bisphenol S, bisphenol F and bisphenol AF induce oxidative stress and biomacromolecular damage in human granulosa KGN cells[J]. Chemosphere, 2020, 253: 126707.
|
| [8] |
WANG B S, HAN J, ELISSEEFF J H, et al. The senescence-associated secretory phenotype and its physiological and pathological implications[J]. Nat Rev Mol Cell Biol, 2024, 25(12): 958-978.
|
| [9] |
SELVARANI R, MOHAMMED S, RICHARDSON A. Effect of rapamycin on aging and age-related diseases-past and future[J]. Geroscience, 2021, 43(3): 1135-1158.
|
| [10] |
LI M Y, SHEN H H, CAO X Y, et al. Targeting a mTOR/autophagy axis: a double-edged sword of rapamycin in spontaneous miscarriage[J]. Biomed Pharmacother, 2024, 177: 116976.
|
| [11] |
SHATROVA A, BUROVA E, PUGOVKINA N, et al. Monovalent ions and stress-induced senescence in human mesenchymal endometrial stem/stromal cells[J]. Sci Rep, 2022, 12(1): 11194.
|
| [12] |
LEONE ROBERTI MAGGIORE U, CHIAPPA V, CECCARONI M, et al. Epidemiology of infertility in women with endometriosis[J]. Best Pract Res Clin Obstet Gynaecol, 2024, 92: 102454.
|
| [13] |
WANG Y D, TANG Z H, TENG X X. New advances in the treatment of thin endometrium[J]. Front Endocrinol, 2024, 15: 1269382.
|
| [14] |
WENG Z J, WANG Y G, OUCHI T, et al. Mesenchymal stem/stromal cell senescence: hallmarks, mechanisms, and combating strategies[J]. Stem Cells Transl Med, 2022, 11(4): 356-371.
|
| [15] |
STEIN G H, DRULLINGER L F, SOULARD A, et al. Differential roles for cyclin-dependent kinase inhibitors p21 and p16 in the mechanisms of senescence and differentiation in human fibroblasts[J]. Mol Cell Biol, 1999, 19(3): 2109-2117.
|
| [16] |
BUROVA E, BORODKINA A, SHATROVA A, et al. Sublethal oxidative stress induces the premature senescence of human mesenchymal stem cells derived from endometrium[J]. Oxid Med Cell Longev, 2013, 2013: 474931.
|
| [17] |
WU X R, LI S Q, NI Y Y, et al. Maternal BPAF exposure impaired synaptic development and caused behavior abnormality in offspring[J]. Ecotoxicol Environ Saf, 2023, 256: 114859.
|
| [18] |
CHEN D, KANNAN K, TAN H L, et al. Bisphenol analogues other than BPA: environmental occurrence, human exposure, and toxicity-a review[J]. Environ Sci Technol, 2016, 50(11): 5438-5453.
|
| [19] |
ZHANG Y Y, LI T J, PAN C Y, et al. Intergenerational toxic effects of parental exposure to bisphenol AF on offspring and epigenetic modulations in zebrafish[J]. Sci Total Environ, 2022, 823: 153714.
|
| [20] |
LIU X X, WANG Z X, LIU F J. Chronic exposure of BPA impairs male germ cell proliferation and induces lower sperm quality in male mice[J]. Chemosphere, 2021, 262: 127880.
|
| [21] |
FAN H J, JIANG L H, LEE Y L, et al. Bisphenol compounds regulate decidualized stromal cells in modulating trophoblastic spheroid outgrowth and invasion in vitro [J]. Biol Reprod, 2020, 102(3): 693-704.
|
| [22] |
SIRACUSA J S, YIN L, MEASEL E, et al. Effects of bisphenol A and its analogs on reproductive health: a mini review[J]. Reprod Toxicol, 2018, 79: 96-123.
|
| [23] |
FERREIRA R, AMARAL C, CORREIA-DA-SILVA G, et al. Bisphenols A, F, S and AF trigger apoptosis and/or endoplasmic reticulum stress in human endometrial stromal cells[J]. Toxicology, 2022, 478: 153282.
|
| [24] |
MA Y, QI M, AN Y, et al. Autophagy controls mesenchymal stem cell properties and senescence during bone aging[J]. Aging Cell, 2018, 17(1): e12709.
|
| [25] |
GARCÍA-PRAT L, MARTÍNEZ-VICENTE M, PERDIGUERO E, et al. Autophagy maintains stemness by preventing senescence[J]. Nature, 2016, 529(7584): 37-42.
|
| [26] |
KANG C, ELLEDGE S J. How autophagy both activates and inhibits cellular senescence[J]. Autophagy, 2016, 12(5): 898-899.
|
| [27] |
QI Z Y, YANG W Q, XUE B B, et al. ROS-mediated lysosomal membrane permeabilization and autophagy inhibition regulate bleomycin-induced cellular senescence[J]. Autophagy, 2024, 20(9): 2000-2016.
|
| [28] |
LYU W P, LI Q H, WANG Y M, et al. Computational design of binder as the LC3-p62 protein-protein interaction[J]. Bioorg Chem, 2021, 115: 105241.
|
| [29] |
KABEYA Y, MIZUSHIMA N, UENO T, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing[J]. EMBO J, 2000, 19(21): 5720-5728.
|
 )
)