吉林大学学报(医学版) ›› 2026, Vol. 52 ›› Issue (1): 162-170.doi: 10.13481/j.1671-587X.20260117
凉膈散对LPS诱导的急性肺损伤的保护作用及其机制
于鑫1,董铮2,张引1,蔡相龙1,刘亮亮1,孙娜1,牛文博3,杨磊4,杨文杰5( )
)
- 1.武警特色医学中心综合重症医学科,天津 300162
2.武警天津总队医院内二科,天津 300162
3.锦州海关综合技术服务中心,辽宁 锦州 121013
4.天津市南开医院急腹症研究所,天津 300100
5.天津市第一中心医院感染科,天津 300190
Protective effect of Liangge powder on LPS-induced acute lung injury and its mechanism
Xin YU1,Zheng DONG2,Yin ZHANG1,Xianglong CAI1,Liangliang LIU1,Na SUN1,Wenbo NIU3,Lei YANG4,Wenjie YANG5( )
)
- 1.Department of Respiratory and Critical Care Medicine,Characteristic Medical Center of Chinese People’s Armed Police Forces,Tianjin 300162,China
2.Department of Internal Medicine,Tianjin Armed Police Corps Hospital,Tianjin 300162,China
3.Comprehensive Technical Service Center of Jinzhou Customs,Jinzhou 121013,China
4.Tianjin Key Laboratory of Acute Abdomen Disease Associated Organ Injury and ITCWM Repair,Tianjin Nankai Hospital,Tianjin,300100,China
5.Department of Infection,First Central Hospital,Tianjin City,Tianjin 300190,China
摘要:
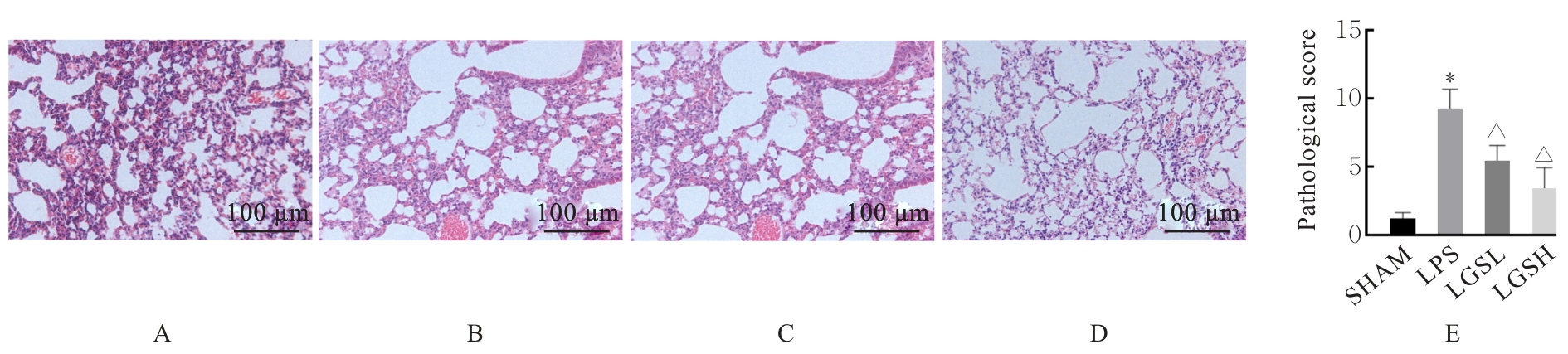
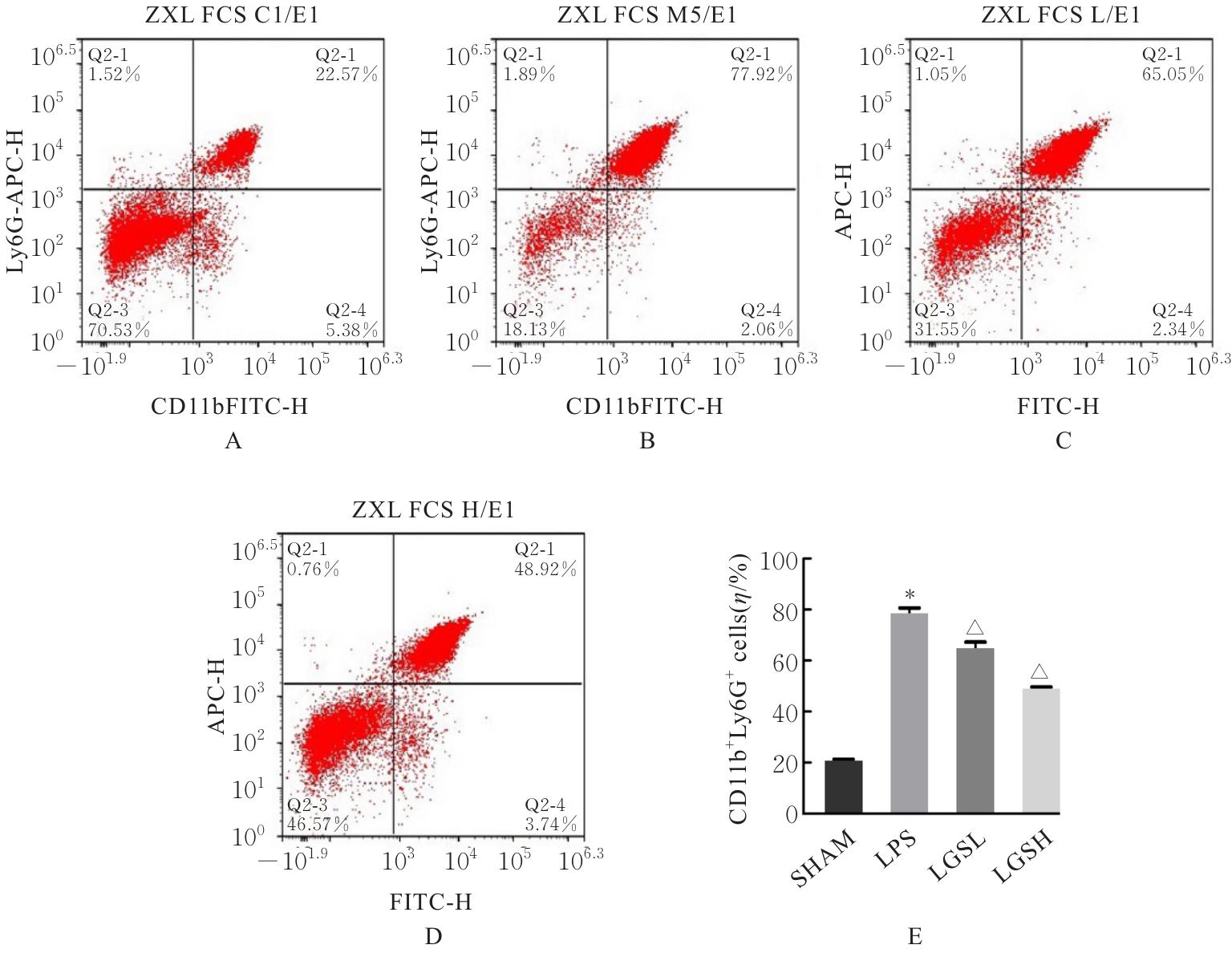
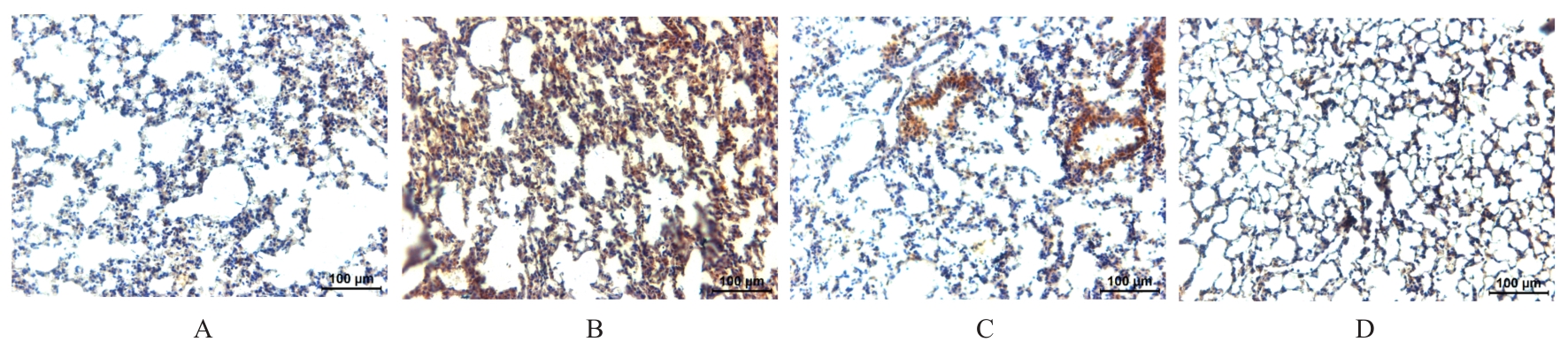
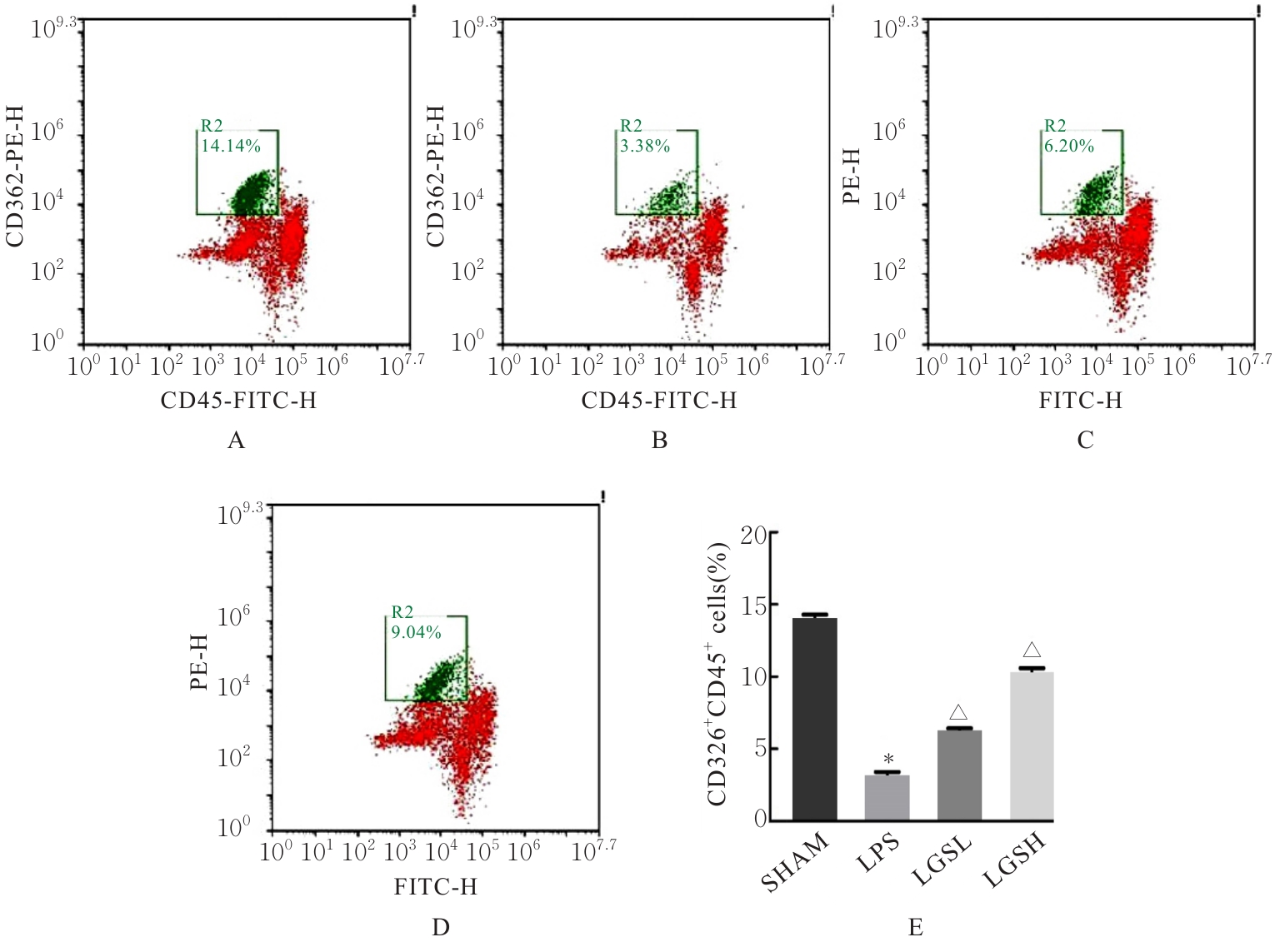
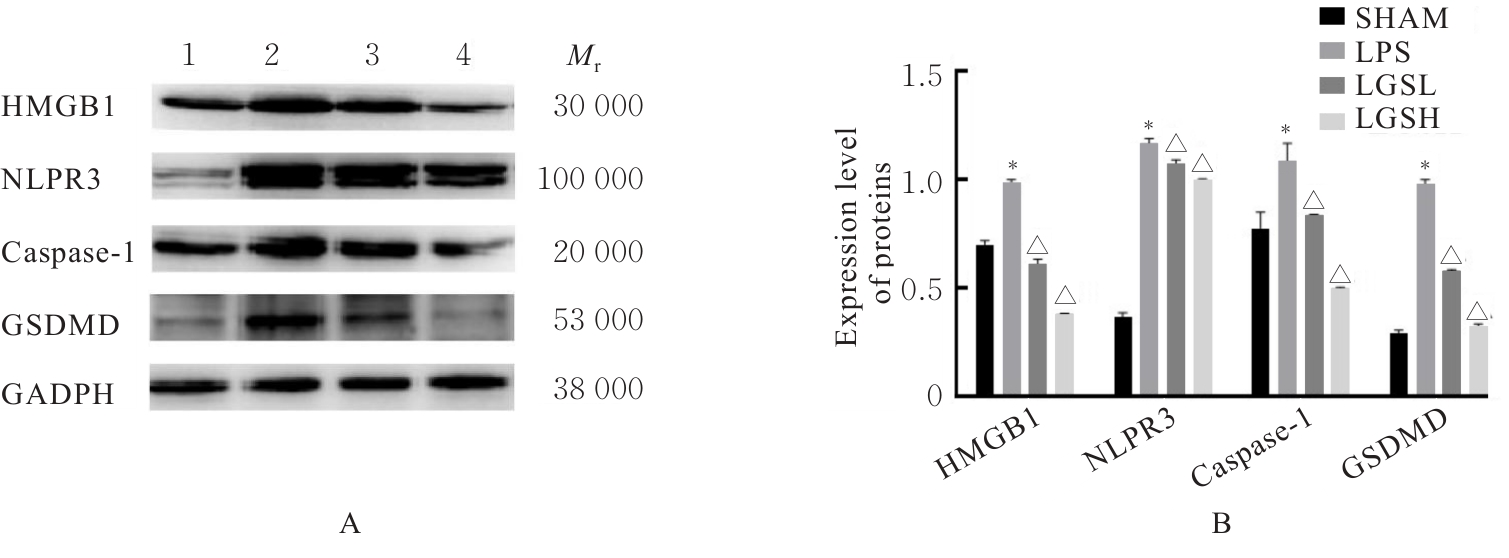
目的 探讨凉膈散(LGS)对脂多糖(LPS)诱导的急性肺损伤(ALI)的保护作用,并阐明其作用机制。 方法 将40只雄性的C57BL/6小鼠随机分为假手术组(SHMA组)、模型组(LPS组)、低剂量LGS组(LGSL组,8.58 g·kg-1 LGS)和高剂量LGS组(LGSH组,17.16 g·kg-1 LGS),每组10只。LPS组、LGSL组和LSGH组小鼠采用气管滴注LPS诱导建立ALI模型,造模24 h后取材。采用HE染色法观察各组小鼠肺组织病理形态表现并进行病理评分,流式细胞术检测各组小鼠肺组织中中性粒细胞和肺泡上皮细胞百分率,免疫组织化学染色法检测各组小鼠肺组织中髓过氧化物酶(MPO)水平,酶联免疫吸附试验(ELISA)法检测各组小鼠肺泡灌洗液中相关炎性介质白细胞介素18(IL-18)、白细胞介素1β(IL-1β)和白蛋白水平,Western blotting法检测各组小鼠肺组织中高迁移率族蛋白B1(HMGB1)、NLR家族Pyrin域蛋白3(NLRP3)、含半胱氨酸的天冬氨酸蛋白水解酶1(Caspase-1)和消皮素D(GSDMD)蛋白表达水平。 结果 HE染色,与SHMA组比较,LPS组小鼠肺组织病理损伤加重,病理评分明显升高(P<0.05);与LPS组比较,LGSL组和LGSH组小鼠肺组织病理学评分明显降低(P<0.05)。流式细胞术,与SHMA组比较,LPS组小鼠肺组织中中性粒细胞百分率明显升高(P<0.05),肺泡上皮细胞百分率明显降低(P<0.05);与LPS组比较,LGSL组和LGSH组小鼠肺组织中中性粒细胞百分率明显降低(P<0.05),肺泡上皮细胞百分率明显升高(P<0.05)。免疫组织化学染色法,与SHMA组比较,LPS组小鼠肺组织中MPO水平明显升高(P<0.05);与LPS组比较,LGSL组和LGSH组小鼠肺组织中MPO水平明显降低(P<0.05)。ELISA法,与SHMA组比较,LPS组小鼠肺泡灌洗液中IL-18、IL-1β和白蛋白水平明显升高(P<0.05);与LPS组比较,LGSL组和LGSH组小鼠肺泡灌洗液中IL-18、IL-1β和白蛋白水平明显降低(P<0.05)。Western blotting法,与SHMA组比较,LPS组小鼠肺组织中HMGB1、NLRP3、Caspase-1和GSDMD蛋白表达水平均明显升高(P<0.05);与LPS组比较,LGSL组和LGSH组小鼠肺组织中HMGB1、NLRP3、Caspase-1和GSDMD蛋白表达水平明显降低(P<0.05)。 结论 LGS可通过降低中性粒细胞浸润和上皮细胞焦亡以减轻LPS诱导的病理肺损伤和炎症反应,其机制可能与其通过HMGB1信号通路下调NLRP3、Caspase-1和GSDMD蛋白表达有关。
中图分类号:
- R285.5