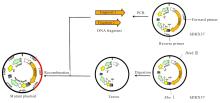
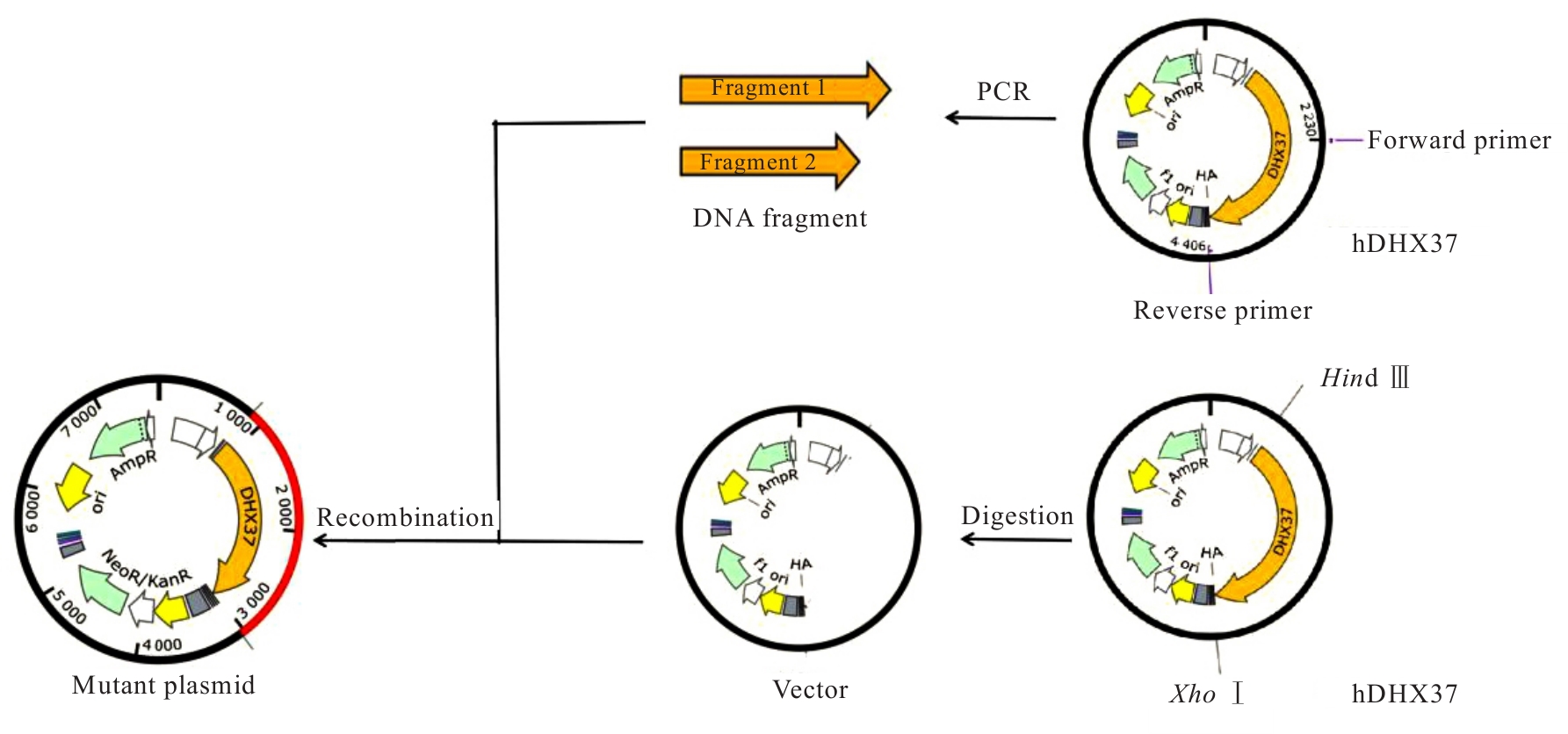
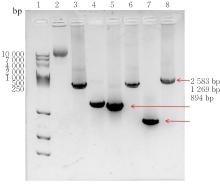
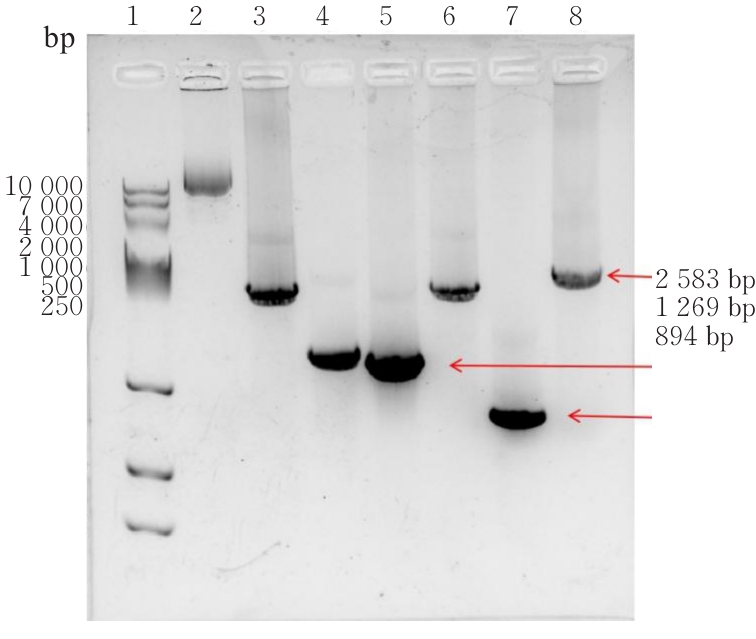
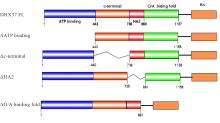
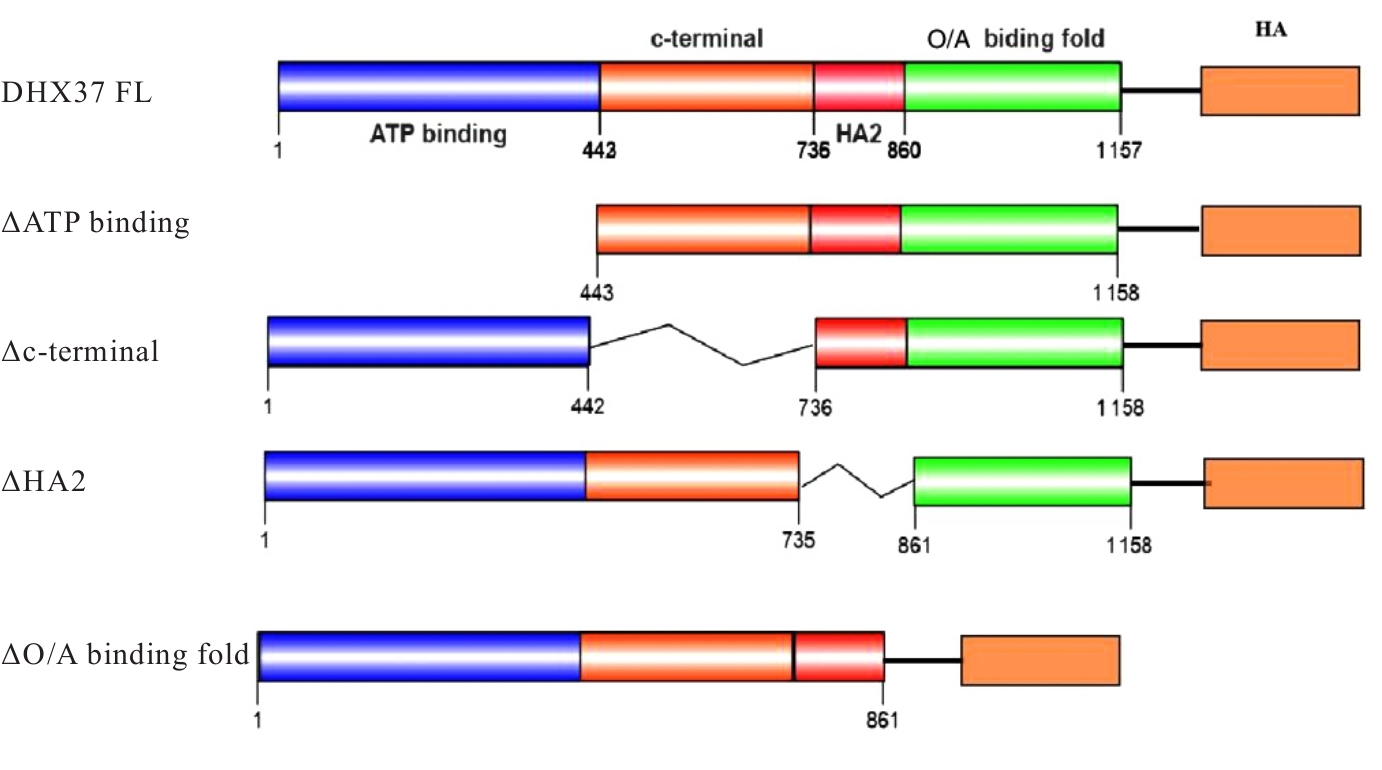
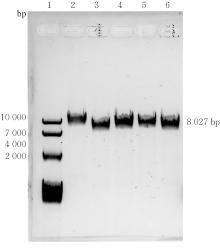
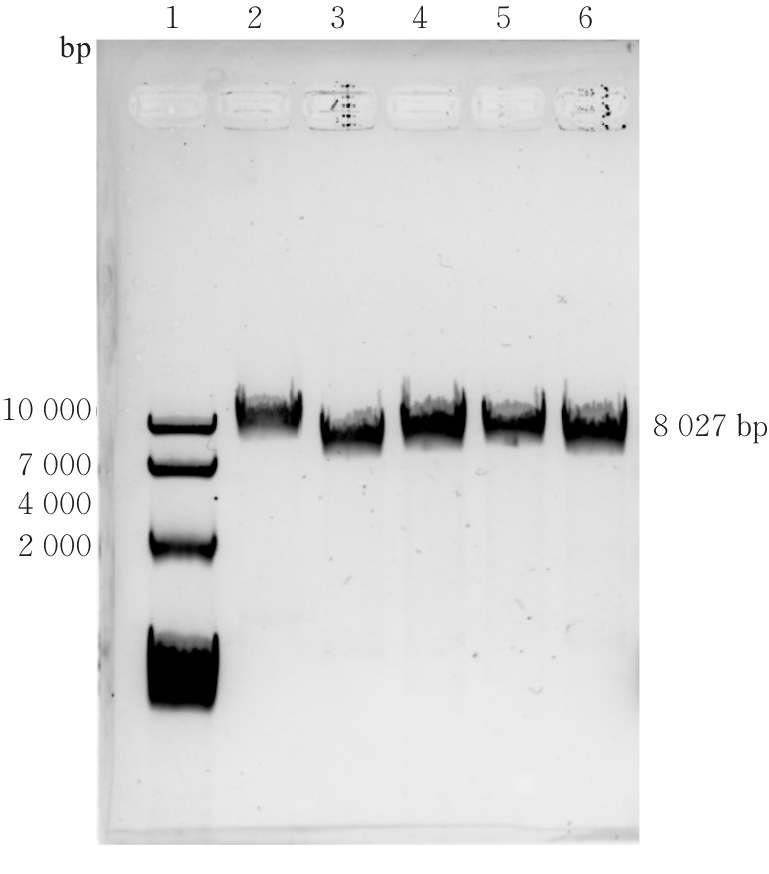
| [1] |
Feifei JIANG,Haoyu LI,Huijian JIA,Dan WANG,Xiaoying ZHAO,Yuefeng WANG,Tong ZHOU,Liyuan SUN.
Establishment and evaluation of detection method of three toxin genes of Bacillus cereus based on multiplex PCR
[J]. Journal of Jilin University(Medicine Edition), 2022, 48(5): 1223-1228.
|
| [2] |
Huijian JIA,Haoyu LI,Dan WANG,Feifei JIANG,Xiaoying ZHAO,Jingyao DONG,Liyuan SUN.
Establishment of method for rapid detection of Bacillus cereus entFMtoxin gene using nucleic acid test strips
[J]. Journal of Jilin University(Medicine Edition), 2022, 48(5): 1333-1340.
|
| [3] |
Zheng GUI,Jingfeng YU,Lan MU.
Detection of DNA in spotted fever group Rickettsia carried by Dermacentor nuttalli in partial areas of Inner Mongolia and its distribution of genotypes
[J]. Journal of Jilin University(Medicine Edition), 2021, 47(1): 210-215.
|
| [4] |
ZHANG Lifang, CAI Lin, WANG Yunhua, ZHANG Keshi, WU Rina.
Analysis on association between selenoprotein P polymorphism and risk of papillary thyriod carcinoma
[J]. Journal of Jilin University(Medicine Edition), 2020, 46(02): 383-388.
|
| [5] |
RAN Nan, MA Mingxing, PANG Zhiqiang, WANG Zeyu, LIU Yue, ZHENG Ruipeng, LU Junying, ZHANG Chao, CHEN Guang, ZHANG Hong, WANG Fang.
Development and evaluation of thrombosis susceptibility gene chip
[J]. Journal of Jilin University(Medicine Edition), 2020, 46(01): 182-187.
|
| [6] |
GAO Lijun, HE Chengyuan, LI Yingnuo, BA Hongyu, LI Zitong, XIA Wei, LI Mingcheng, YUAN Guanxin, ZHANG Lihua, AI Jinxia.
Characteristics and identification of DNA fingerprint of velvet antler and its counterfeits based on duplex PCR technique
[J]. Journal of Jilin University Medicine Edition, 2018, 44(04): 839-844.
|
| [7] |
HUANG Baoliang, DING Chuanbo, WANG Jiaqi, ZHENG Yinan, LIU Wencong, ZHANG Jing, XU Xiaohua.
Anti-fatigue effect of arginylfructosylglucose from red ginseng in mice
[J]. Journal of Jilin University Medicine Edition, 2017, 43(05): 897-902.
|
| [8] |
YANG Zhujun, HOU Xu, YU Wenwen, YAN Kuo, SHEN Yuqin, SUN Xinhua.
Effects of MT01 on expressions of TLR9,TRAF6 and IL6 in periodontal tissue during experimental tooth movement of rats
[J]. Journal of Jilin University Medicine Edition, 2017, 43(04): 752-756.
|
| [9] |
WEN Aiping, WANG Haijiao, JIN Jingji, CAI Yong, ZHU Jihong, CHEN Lunhua.
Expressions of hMOFprotein and mRNA in endometrial cancer tissue and their clinical significances
[J]. Journal of Jilin University Medicine Edition, 2016, 42(06): 1138-1142.
|
| [10] |
LIU Huanxin, ZHANG Guoxiang, GUO Linlang, TAN Songshan.
Expression of miR-139-5p in small cell lung cancer tissue and its clinical significance
[J]. Journal of Jilin University Medicine Edition, 2016, 42(05): 942-948.
|
| [11] |
DONG Zefeng, ZHENG Tianli, FAN Xuejia, Pei Xiaofang.
Epidemic characteristics of rhinovirus in hospitalized children with respiratory syndrome in Chengdu during 2011-2013
[J]. Journal of Jilin University Medicine Edition, 2016, 42(01): 177-181.
|
| [12] |
SUN Xiaohong, FANG Fuyuan, LI Hui, YIN Na, PANG Zuoliang, LI Huiwu.
Expressions of MMP-2 and MMP-9 in tumor tissue of patients with esophageal squamous cell carcinoma and their significances
[J]. Journal of Jilin University Medicine Edition, 2015, 41(04): 814-819.
|
| [13] |
PENG Jing, YUAN Ruili, WANG Xiaoqin, WU Feng, GUO Xuan.
Detection of expression levels of microRNA-181a and microRNA-20a in serum of patients with multiple myeloma and its clinical significance
[J]. Journal of Jilin University Medicine Edition, 2015, 41(03): 625-630.
|
| [14] |
ZHAO Min-xue,LI Bai-wen,ZHU Lin,YE Hong-shan,TIAN Xiao-feng,WEI Liang,CAO Hong.
Detection of miR-21 expression levels in carcinoma tissue and serum of patients with papillary thyroid carcinoma and its significance
[J]. Journal of Jilin University Medicine Edition, 2014, 40(06): 1237-1240.
|
| [15] |
MA Ning,DONG Xiao-yan,JIANG Yan-fang,LIU Meng-meng,LIUZi-ling.
Construction of recombinant adenovirus vector pAV.Ex1d-Hiwi using Gateway technology
[J]. Journal of Jilin University Medicine Edition, 2014, 40(04): 898-903.
|
 )
)