吉林大学学报(医学版) ›› 2026, Vol. 52 ›› Issue (1): 44-55.doi: 10.13481/j.1671-587X.20260106
葛根素对高脂饮食诱导小鼠非酒精性脂肪性肝病的改善作用及其机制
陈思童1,2,杨丹1,3,李庆杰1,3,唐晓雷1,3,王汉1,2,刘扬扬1,4( ),刘铁军1,2(
),刘铁军1,2( )
)
- 1.长春中医药大学中医学院中医学系,吉林 长春 130117
2.长春中医药大学附属医院肝脾胃病科,吉林 长春 130021
3.长春中医药大学附属医院中医药研究中心,吉林 长春 130021
4.长春中医药 大学附属医院治未病科,吉林 长春 130021
Ameliorative effect of puerarin on non-alcoholic fatty liver disease induced by high-fat diet in mice and its mechanism
Sitong CHEN1,2,Dan YANG1,3,Qingjie LI1,3,Xiaolei TANG1,3,Han WANG1,2,Yangyang LIU1,4( ),Tiejun LIU1,2(
),Tiejun LIU1,2( )
)
- 1.Department of Chinese Medicine,School of Chinese Medicine,Changchun University of Chinese Medicine,Changchun 130117,China
2.Department of Gastroenterology,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
3.Research Center of Chinese Traditional Medicine,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
4.Department of Preventive Treatment,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
摘要:
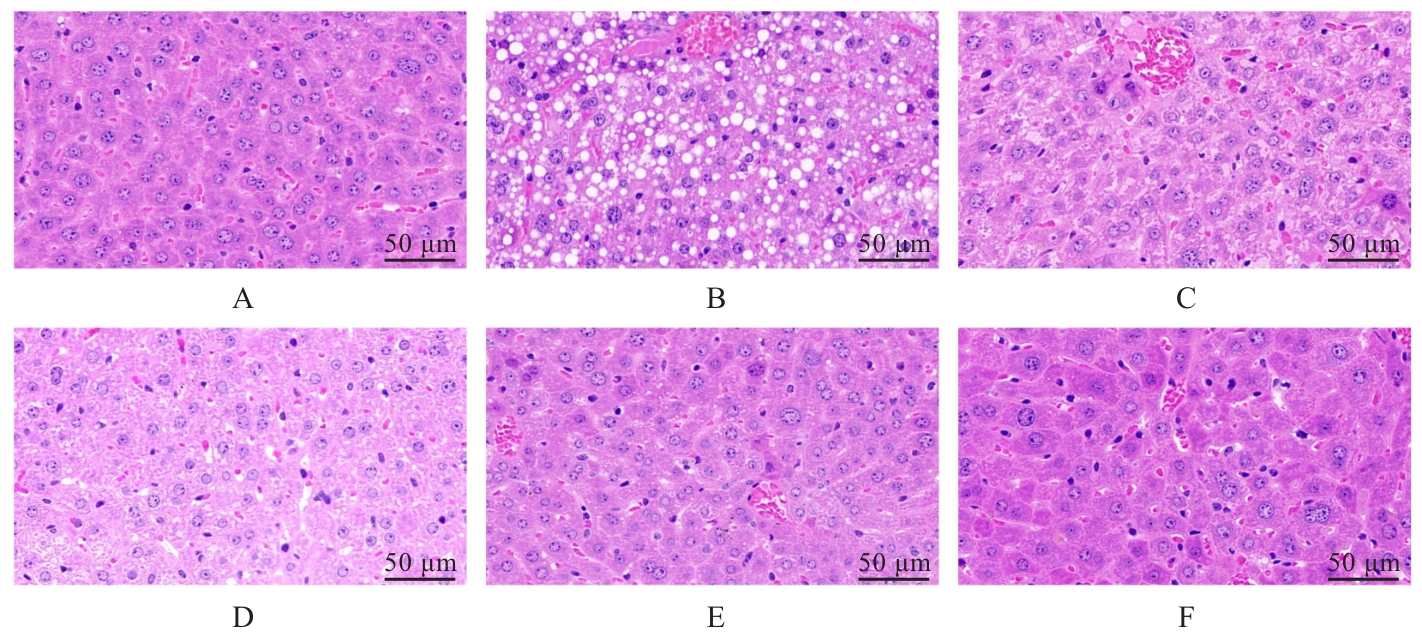
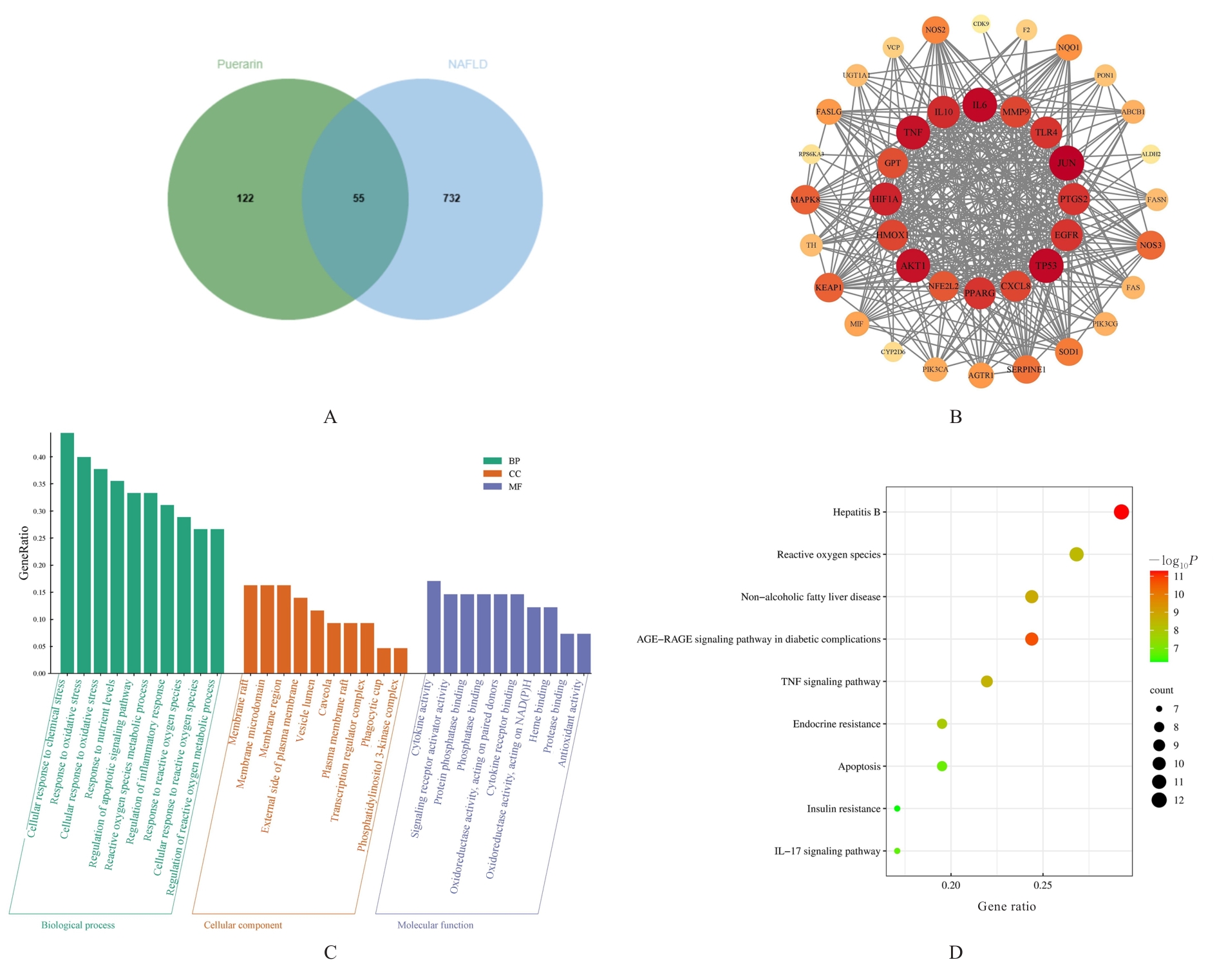
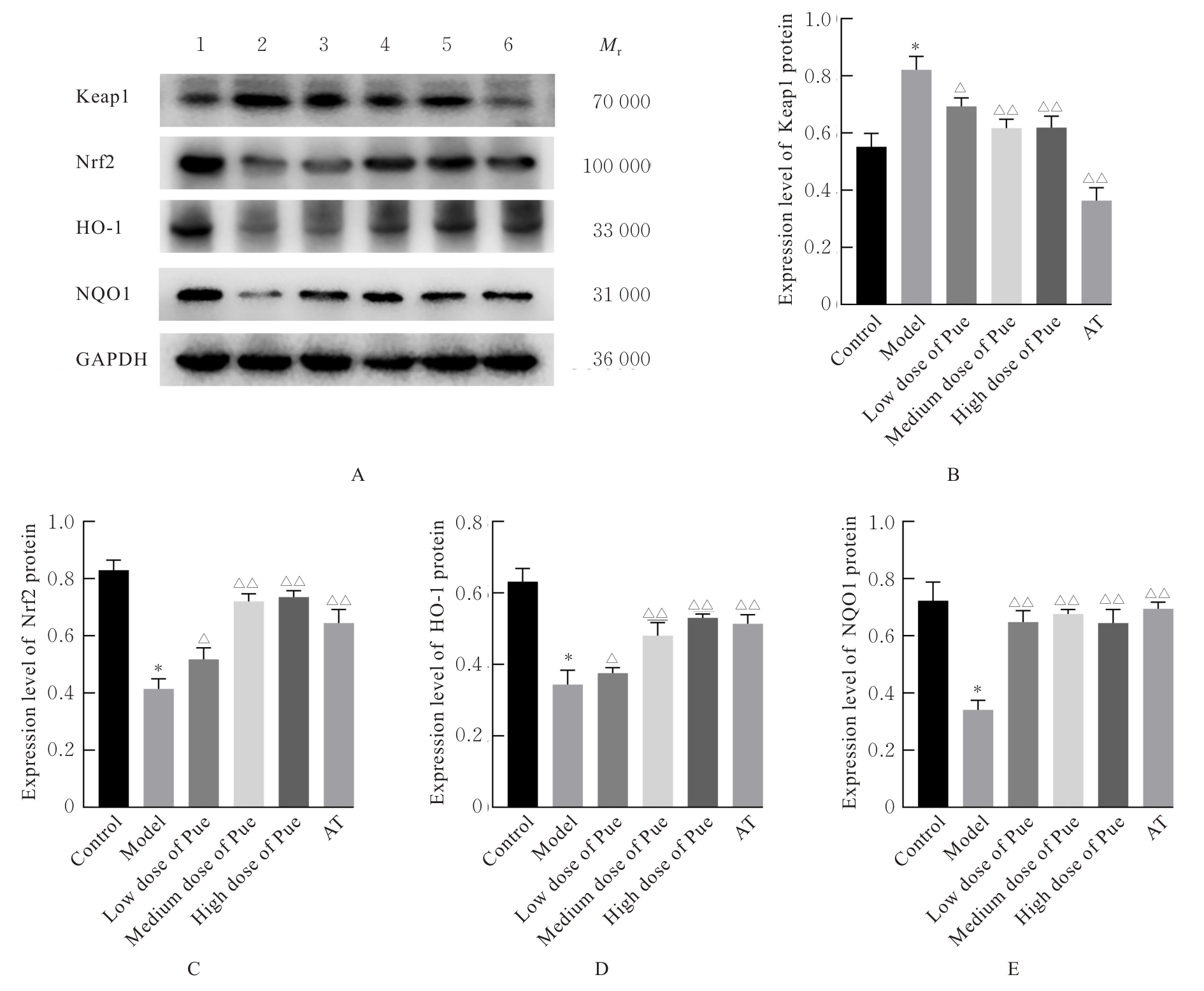
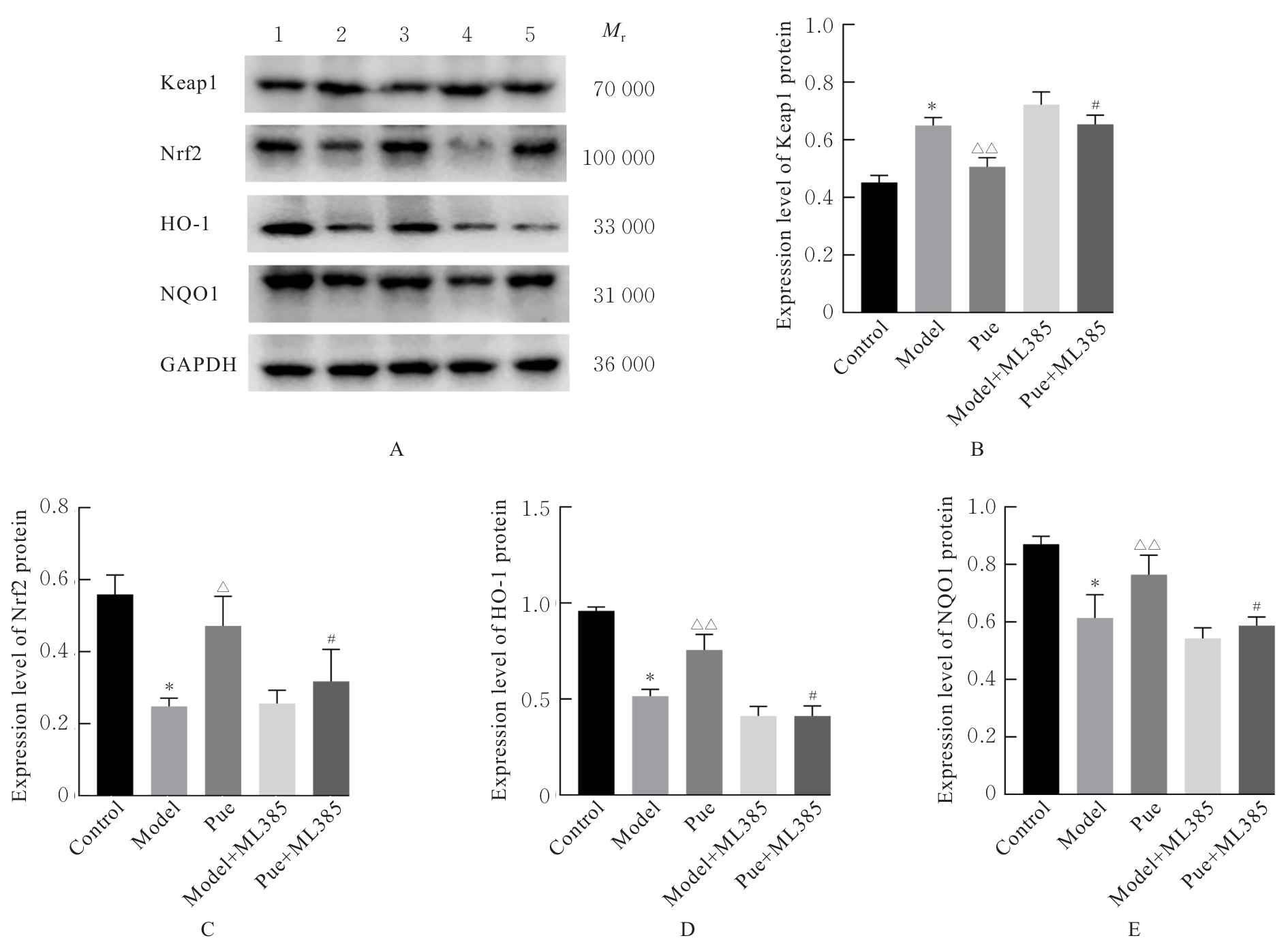
目的 探讨葛根素(Pue)对高脂饮食诱导小鼠非酒精性脂肪性肝病(NAFLD)的改善作用,并阐明其可能的作用机制。 方法 将36只C57BL/6J小鼠随机分为对照组、模型组、低剂量Pue组(20.0 mg·kg-1)、中剂量Pue组(40.0 mg·kg-1)、高剂量Pue组(80.0 mg·kg-1)和阳性药组[阿托伐他汀钙(AT)组](10.0 mg·kg-1 AT),每组6只。除对照组小鼠给予正常饮食外,其余各组小鼠均给予高脂饮食喂养以建立NAFLD模型。测量各组小鼠体质量,并计算肝指数;采用HE染色观察各组小鼠肝组织病理形态表现;采用生化试剂盒测定各组小鼠血清和肝组织中总胆固醇(TC)及甘油三酯(TG)水平,血清丙氨酸氨基转移酶(ALT)和天门冬氨酸氨基转移酶(AST)水平,肝组织中丙二醛(MDA)水平以及超氧化物歧化酶(SOD)和谷胱甘肽过氧化物酶(GSH-Px)活性,并检测肝组织中炎症因子[白细胞介素6(IL-6)、白细胞介素1β(IL-1β)和肿瘤坏死因子α(TNF-α)]水平。基于网络药理学框架,通过整合PubChem和SwissTargetPrediction等数据库预测Pue的潜在治疗靶点,结合GeneCards等平台获取的NAFLD相关疾病靶点构建蛋白质-蛋白质相互作用(PPI)网络,并进行基因本体论(GO)功能和京都基因与基因组百科全书(KEGG)信号通路富集分析。采用Western blotting法检测各组小鼠肝组织中Kelch样ECH关联蛋白1(Keap1)、核因子E2相关因子2(Nrf2)、血红素加氧酶1(HO-1)和醌氧化还原酶1(NQO1)蛋白表达水平。另取30只小鼠分为对照组、模型组、Pue组(80.0 mg·kg-1 Pue灌胃)、模型+Nrf2抑制剂ML385组(30.0 mg·kg-1 ML385腹腔注射)和Pue+Nrf2抑制剂ML385组(80.0 mg·kg-1 Pue灌胃+30 mg·kg-1 ML385腹腔注射),每组6只。再次检测上述氧化应激和炎症指标以及相关蛋白表达情况。 结果 与对照组比较,模型组小鼠体质量和肝指数明显升高(P<0.01);与模型组比较,低、中和高剂量Pue组及AT组小鼠体质量以及肝指数明显降低(P<0.05或P<0.01)。HE染色,与对照组比较,模型组小鼠肝组织出现明显的脂质积累和病理损伤;与模型组比较,低、中和高剂量Pue组及AT组小鼠肝组织的脂滴积累情况以及病理损伤均有不同程度的改善。与对照组比较,模型组小鼠肝组织中TC、TG、IL-6、IL-1β、TNF-α和MDA水平以及血清中TC、TG、ALT和AST水平明显升高(P<0.01),肝组织中SOD和GSH-Px活性明显降低(P<0.01);与模型组比较,低、中和高剂量Pue组及AT组小鼠肝组织中TC、TG、IL-6、IL-1β、TNF-α和MDA水平以及血清中TC、TG、ALT和AST水平明显降低(P<0.05或P<0.01),肝组织中SOD和GSH-Px活性明显升高(P<0.05或P<0.01)。网络药理学分析筛选获得Pue治疗NAFLD的核心靶点包括Nrf2、TNF和IL-6等,预测Pue可能通过调控氧化应激、炎症和凋亡等途径发挥作用。Western blotting法,与对照组比较,模型组小鼠肝组织中Keap1蛋白表达水平升高(P<0.01),Nrf2、HO-1和NQO1蛋白表达水平均降低(P<0.01);与模型组比较,低、中和高剂量Pue组及AT组小鼠肝组织中Keap1蛋白表达水平降低(P<0.05或P<0.01),Nrf2、HO-1和NQO1蛋白表达水平均升高(P<0.05或P<0.01)。Nrf2抑制剂ML385干预后,与Pue组比较,Pue+ML385组小鼠肝组织中IL-6、IL-1β、TNF-α和MDA水平均明显升高(P<0.05),SOD和GSH-Px活性均明显降低(P<0.05或P<0.01),肝组织中Keap1蛋白表达水平升高(P<0.01),Nrf2、HO-1和NQO1蛋白表达水平均明显降低(P<0.01)。 结论 Pue能够改善NAFLD小鼠的脂质代谢和肝功能,减轻肝脏损伤,提高肝组织抗氧化能力,并减少炎症因子释放,其机制可能与激活Keap1/Nrf2/HO-1信号通路有关。
中图分类号:
- R285.5