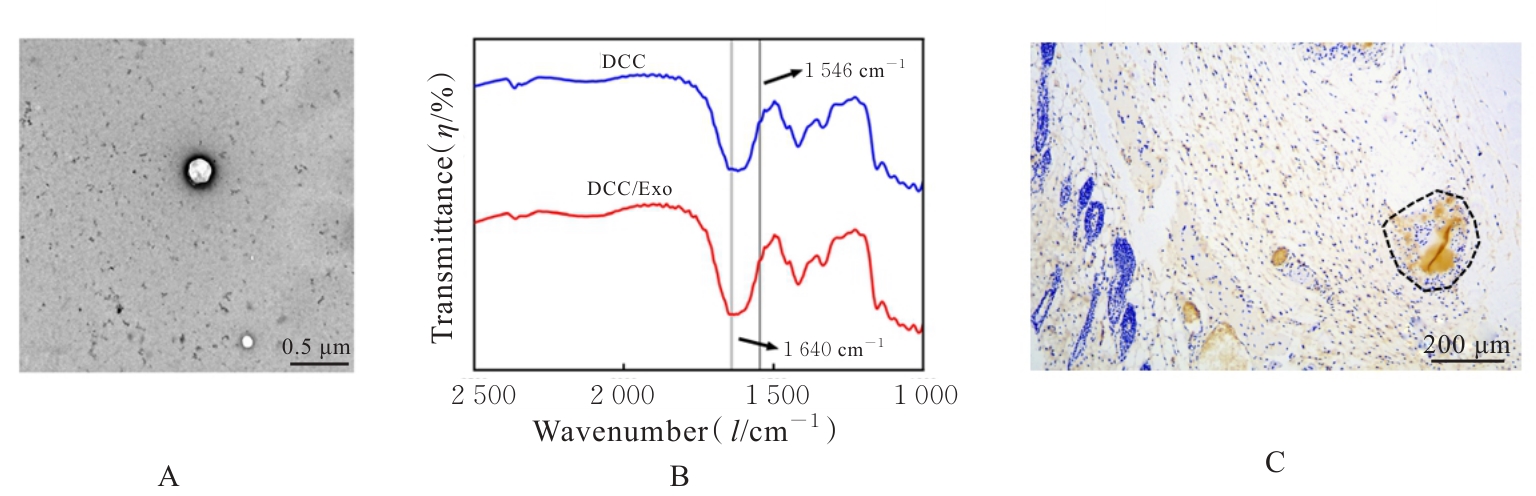
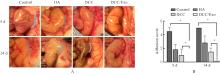
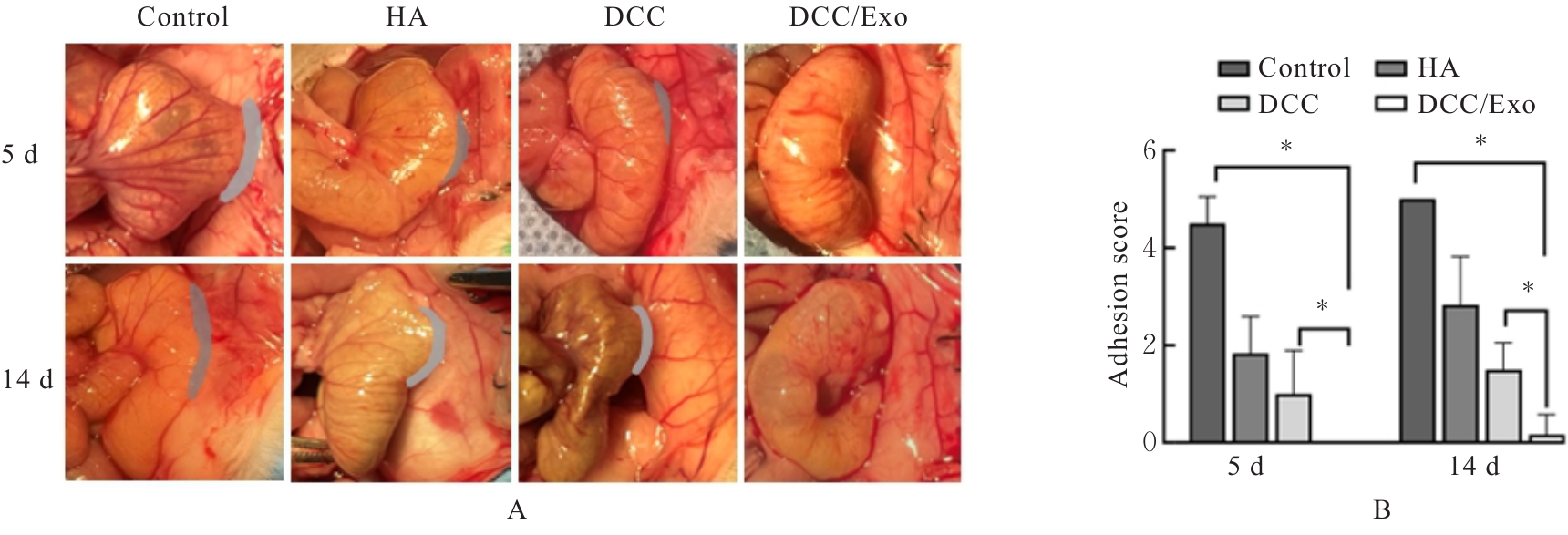
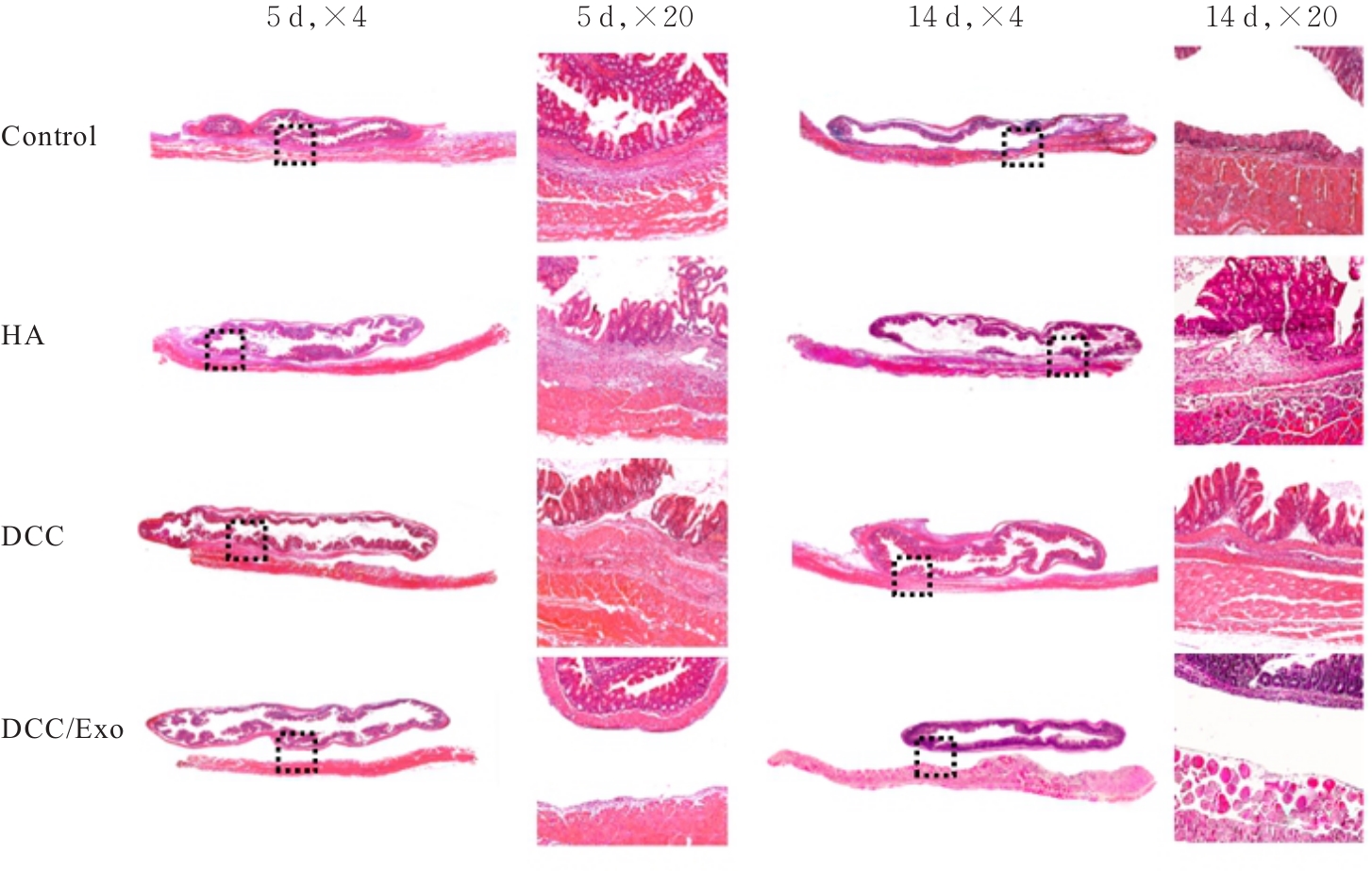
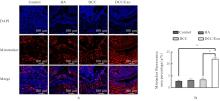
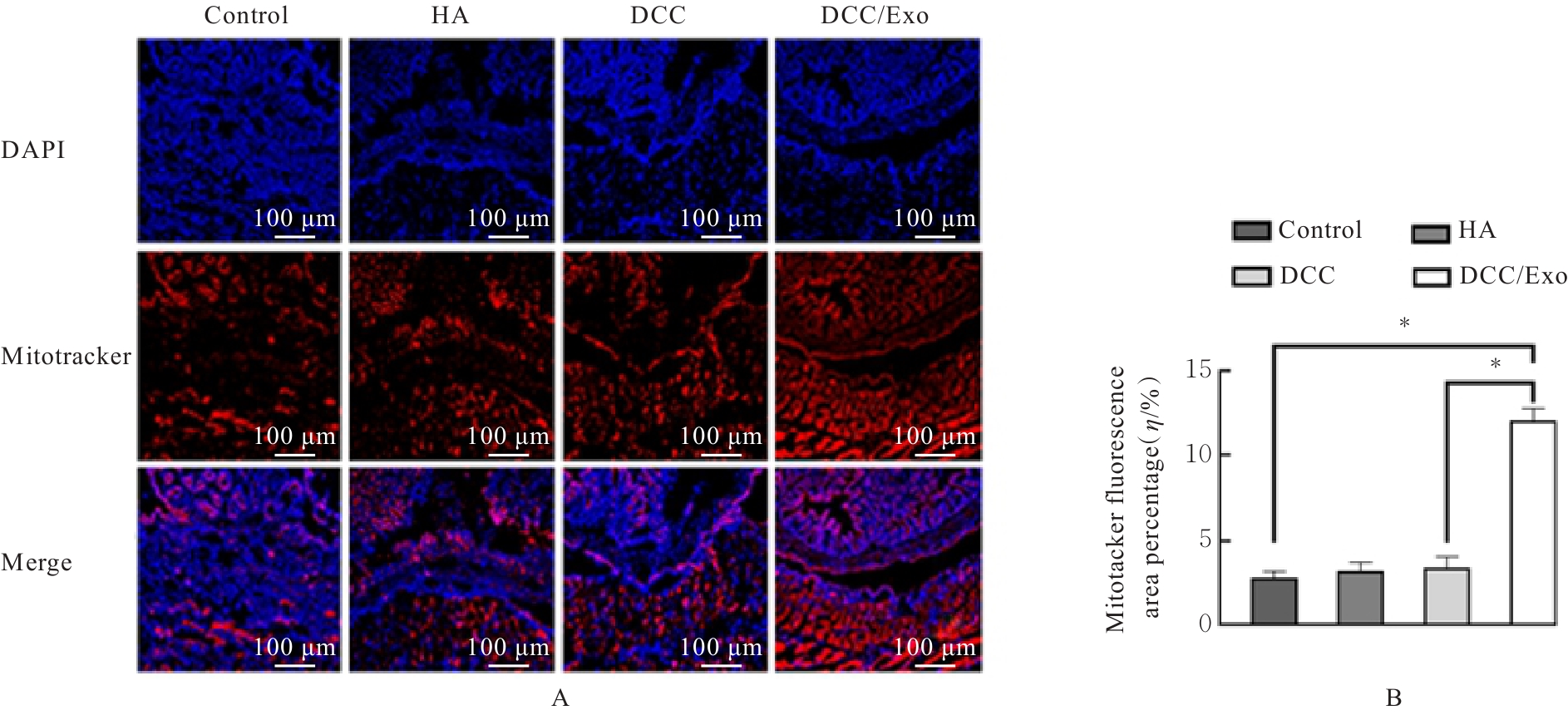
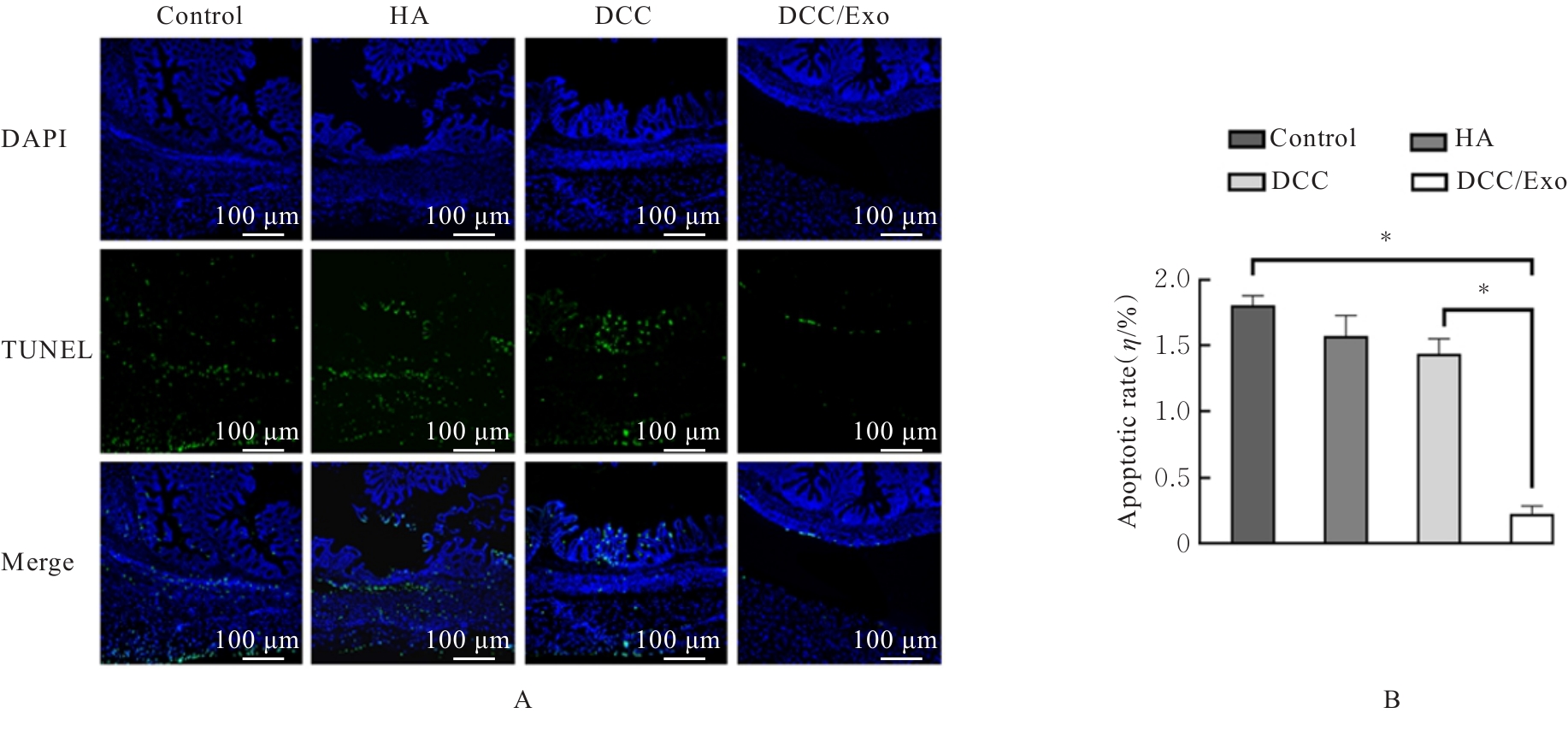
| [1] |
VAKILIAN S, AL-HASHMI S, AL-KINDI J, et al. Avastin-loaded 3D-printed alginate scaffold as an effective antiadhesive barrier to prevent postsurgical adhesion bands formation[J]. Macromol Biosci, 2024, 24(6): e2300530.
|
| [2] |
LI X Y, CAI J, DUAN X M, et al. Injectable polyamide-amine dendrimer-crosslinked meloxicam-containing poly-γ-glutamic acid hydrogel for prevention of postoperative tissue adhesion through inhibiting inflammatory responses and balancing the fibrinolytic system[J]. J Colloid Interface Sci, 2024, 670: 486-498.
|
| [3] |
MA Y Y, SUN T H, REN K J, et al. Plasma-activated solutions prevent peritoneal adhesion formation by regulating eNOS expression in mesothelial cells[J]. J Adv Res, 2025, 78: 555-571.
|
| [4] |
CHEN Y, WANG X, TAO S, et al. Research advances in smart responsive-hydrogel dressings with potential clinical diabetic wound healing properties[J]. Mil Med Res, 2023, 10(1): 37.
|
| [5] |
江俊杰, 武 皓, 何 康, 等. 人参多肽温敏水凝胶对大鼠热源性皮肤损伤的修复作用及其机制[J]. 吉林大学学报(医学版), 2025, 51(2): 360-369.
|
| [6] |
MONDAL J, PILLARISETTI S, JUNNUTHULA V, et al. Hybrid exosomes, exosome-like nanovesicles and engineered exosomes for therapeutic applications[J]. J Control Release, 2023, 353: 1127-1149.
|
| [7] |
TAN X, ZHANG J, HENG Y Y, et al. Locally delivered hydrogels with controlled release of nanoscale exosomes promote cardiac repair after myocardial infarction[J]. J Control Release, 2024, 368: 303-317.
|
| [8] |
YU Q Y, SUN H, YUE Z W, et al. Zwitterionic polysaccharide-based hydrogel dressing as a stem cell carrier to accelerate burn wound healing[J]. Adv Healthc Mater, 2023, 12(7): e2202309.
|
| [9] |
FAN M H, PI J K, ZOU C Y, et al. Hydrogel-exosome system in tissue engineering: a promising therapeutic strategy[J]. Bioact Mater, 2024, 38: 1-30.
|
| [10] |
ZHANG Z Q, YIN C, SONG X W, et al. A self-fused peptide-loaded hydrogel with injectability and tissue-adhesiveness for preventing postoperative peritoneal adhesions[J]. Mater Today Bio, 2024, 28: 101205.
|
| [11] |
KRISHNAN M A, ALIMI O A, KUSS M, et al. A dual-layer hydrogel barrier integrating bio-adhesive and anti-adhesive properties prevents postoperative abdominal adhesions[J]. Adv Healthc Mater, 2025, 14(11): 2405238.
|
| [12] |
ZHANG Y H, YU L, QIU R J, et al. 3D hypoxia-mimicking and anti-synechia hydrogel enabling promoted neovascularization for renal injury repair and regeneration[J]. Mater Today Bio, 2023, 21: 100694.
|
| [13] |
FOSTER D S, GUO J L, MEANY E, et al. Postoperative adhesions are abrogated by a sustained-release anti-JUN therapeutic in preclinical models[J]. Sci Transl Med, 2025, 17(789): eadp9957.
|
| [14] |
LI X M, GUO Y F, WANG Z Y, et al. Platelet-rich fibrin promotes mesothelial cell proliferation and peritoneal repair by up-regulating calretinin to prevent postoperative intestinal adhesion[J]. Int J Med Sci, 2025, 22(6): 1254-1268.
|
| [15] |
JIANG L J, YAO F L, ZHANG E S, et al. Combined treatment of xyloglucan derivative hydrogel and anti-C5a receptor antibody in preventing peritoneal adhesion[J]. Acta Biomater, 2022, 151: 163-173.
|
| [16] |
GONG P, REN L L, GAO X H, et al. A novel barrier membrane with long-term ROS scavenging function for complete prevention of postoperative adhesion[J]. Mater Des, 2024, 238: 112691.
|
| [17] |
MASOLA V, BONOMINI M, BORRELLI S, et al. Fibrosis of peritoneal membrane as target of new therapies in peritoneal dialysis[J]. Int J Mol Sci, 2022, 23(9): 4831.
|
| [18] |
LU Y Q, ELROD J, HERRMANN M, et al. Neutrophil extracellular traps: a crucial factor in post-surgical abdominal adhesion formation[J]. Cells, 2024, 13(11): 991.
|
| [19] |
HERRO R, GRIMES H L. The diverse roles of neutrophils from protection to pathogenesis[J]. Nat Immunol, 2024, 25(12): 2209-2219.
|
| [20] |
CHEN J M, AN X Y, XU L, et al. Adhesive nanoparticle-in-microgel system with ROS scavenging capability and hemostatic activity for postoperative adhesion prevention[J]. Small, 2024, 20(27): e2306598.
|
| [21] |
REN Y W, LI G, LI E M, et al. Luteolin blocks the ROS/PI3K/AKT pathway to inhibit mesothelial-mesenchymal transition and reduce abdominal adhesions[J]. Eur J Pharmacol, 2024, 964: 176272.
|
| [22] |
LI G, REN Y W, LI E M, et al. Quercetin inhibits mesothelial-mesenchymal transition and alleviates postoperative peritoneal adhesions by blocking the TGF-β1/PI3K/AKT pathway[J]. J Ethnopharmacol, 2024, 319(Pt 2): 117242.
|
| [23] |
SANDOVAL P, JIMÉNEZ-HEFFERNAN J A, GUERRA-AZCONA G, et al. Mesothelial-to-mesenchymal transition in the pathogenesis of post-surgical peritoneal adhesions[J]. J Pathol, 2016, 239(1): 48-59.
|
| [24] |
WANG R P, GUO T K, LI J L. Mechanisms of peritoneal mesothelial cells in peritoneal adhesion[J]. Biomolecules, 2022, 12(10): 1498.
|
| [25] |
WANG Y, SHI Y F, TAO M, et al. Peritoneal fibrosis and epigenetic modulation[J]. Perit Dial Int, 2021, 41(2): 168-178.
|
 )
)