Journal of Jilin University(Medicine Edition) ›› 2026, Vol. 52 ›› Issue (1): 44-55.doi: 10.13481/j.1671-587X.20260106
• Research in basic medicine • Previous Articles Next Articles
Ameliorative effect of puerarin on non-alcoholic fatty liver disease induced by high-fat diet in mice and its mechanism
Sitong CHEN1,2,Dan YANG1,3,Qingjie LI1,3,Xiaolei TANG1,3,Han WANG1,2,Yangyang LIU1,4( ),Tiejun LIU1,2(
),Tiejun LIU1,2( )
)
- 1.Department of Chinese Medicine,School of Chinese Medicine,Changchun University of Chinese Medicine,Changchun 130117,China
2.Department of Gastroenterology,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
3.Research Center of Chinese Traditional Medicine,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
4.Department of Preventive Treatment,Affiliated Hospital,Changchun University of Chinese Medicine,Changchun 130021,China
-
Received:2025-03-09Accepted:2025-04-25Online:2026-01-28Published:2026-02-24 -
Contact:Yangyang LIU,Tiejun LIU E-mail:996439299@qq.com;liutiejun699@163.com
CLC Number:
- R285.5
Cite this article
Sitong CHEN,Dan YANG,Qingjie LI,Xiaolei TANG,Han WANG,Yangyang LIU,Tiejun LIU. Ameliorative effect of puerarin on non-alcoholic fatty liver disease induced by high-fat diet in mice and its mechanism[J].Journal of Jilin University(Medicine Edition), 2026, 52(1): 44-55.
share this article
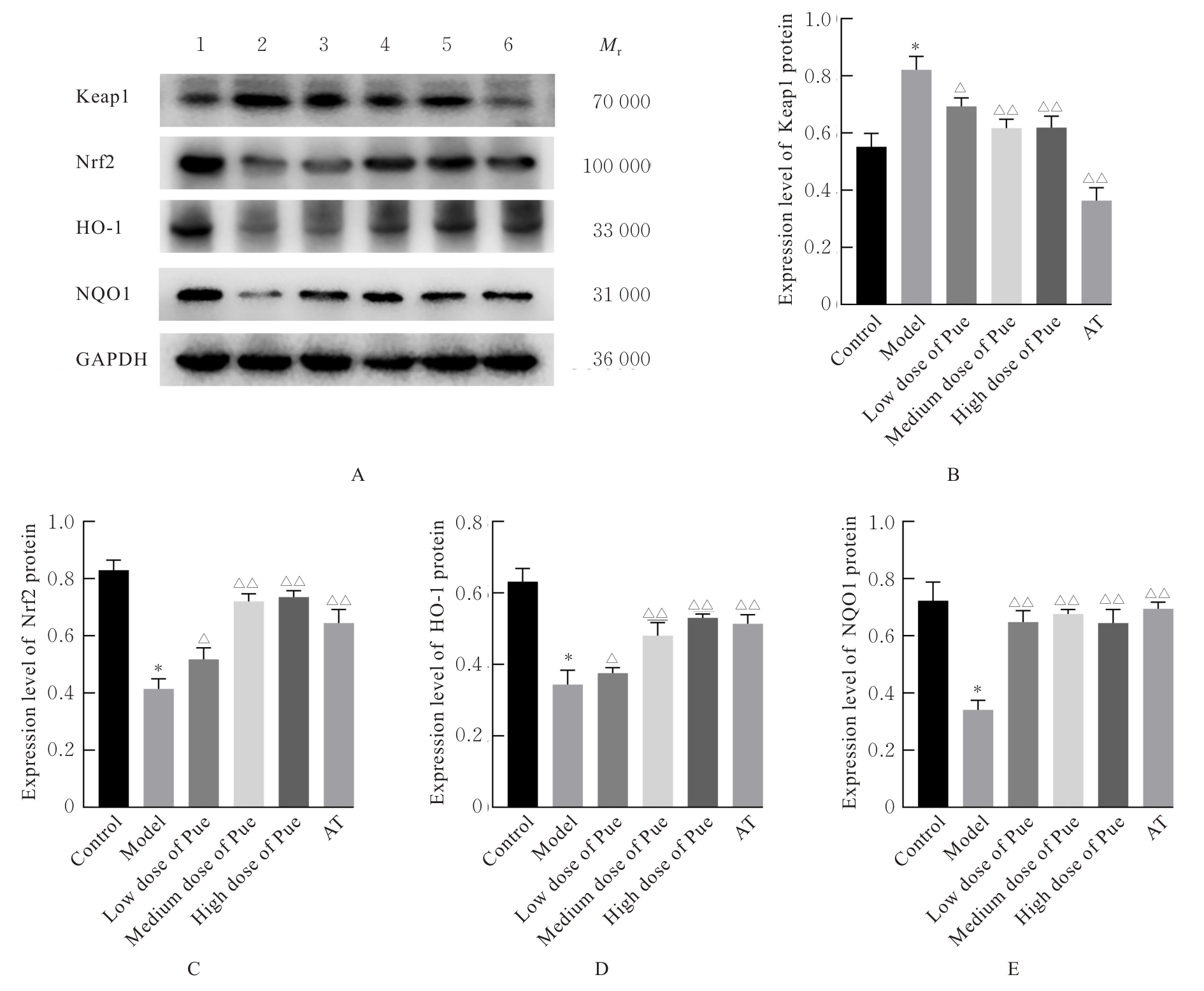
Tab.2
Levels of TC and TG in liver tissue and serum of mice in various groups"
| Group | TC level in liver tissue [mB/(mmol·g-1)] | TG level in liver tissue [mB/(mmol·g-1)] | TC level in serum [cB/(mmol·L-1)] | TG level in serum [cB/(mmol·L-1)] |
|---|---|---|---|---|
| Control | 0.29±0.05 | 0.55±0.05 | 1.52±0.08 | 0.63±0.05 |
| Model | 0.73±0.06* | 1.21±0.05* | 5.95±0.11* | 1.70±0.08* |
| Pue | ||||
| Low dose | 0.60±0.07△ | 1.09±0.06△ | 5.36±0.56△ | 1.49±0.10△ |
| Medium dose | 0.59±0.06△ | 0.82±0.08△△ | 3.46±0.32△△ | 1.06±0.10△△ |
| High dose | 0.53±0.10△△ | 0.67±0.05△△ | 2.84±0.27△△ | 0.86±0.06△△ |
| AT | 0.37±0.08△△ | 0.65±0.06△△ | 2.70±0.40△△ | 0.78±0.06△△ |
Tab.4
Levels of MDA and activities of SOD and GSH-Px in liver tissue of mice in various groups"
| Group | MDA [mB/(μmol·g-1)] | SOD [λB/(U·mg-1)] | GSH-Px [λB/(U·mg-1)] |
|---|---|---|---|
| Control | 1.36±0.08 | 140.48±7.39 | 126.38±6.54 |
| Model | 2.66±0.23* | 57.37±6.94* | 54.01±5.36* |
| Pue | |||
| Low dose | 2.33±0.26△ | 72.80±4.92△ | 67.12±9.16△ |
| Medium dose | 2.00±0.12△△ | 89.46±6.29△△ | 92.87±5.81△△ |
| High dose | 1.87±0.11△△ | 106.12±9.04△△ | 106.02±6.96△△ |
| AT | 1.68±0.18△△ | 120.15±9.42△△ | 109.61±7.18△△ |
Tab.5
Levels of IL-6, IL-1β, and TNF-α in liver tissue of mice in various groups [n=6, x±s,ρB/(ng·L-1)]"
| Group | IL-6 | IL-1β | TNF-α |
|---|---|---|---|
| Control | 15.77±1.12 | 12.42±0.72 | 115.76±8.35 |
| Model | 29.69±3.27* | 31.25±4.28* | 195.25±8.99* |
| Pue | |||
| Low dose | 24.59±1.43△ | 26.24±1.34△ | 178.74±6.43△ |
| Medium dose | 23.27±2.80△△ | 25.77±1.72△ | 165.43±8.27△△ |
| High dose | 21.82±2.80△△ | 22.73±2.52△△ | 143.23±7.47△△ |
| AT | 22.11±3.08△△ | 21.48±3.45△△ | 146.31±12.24△△ |
Tab.6
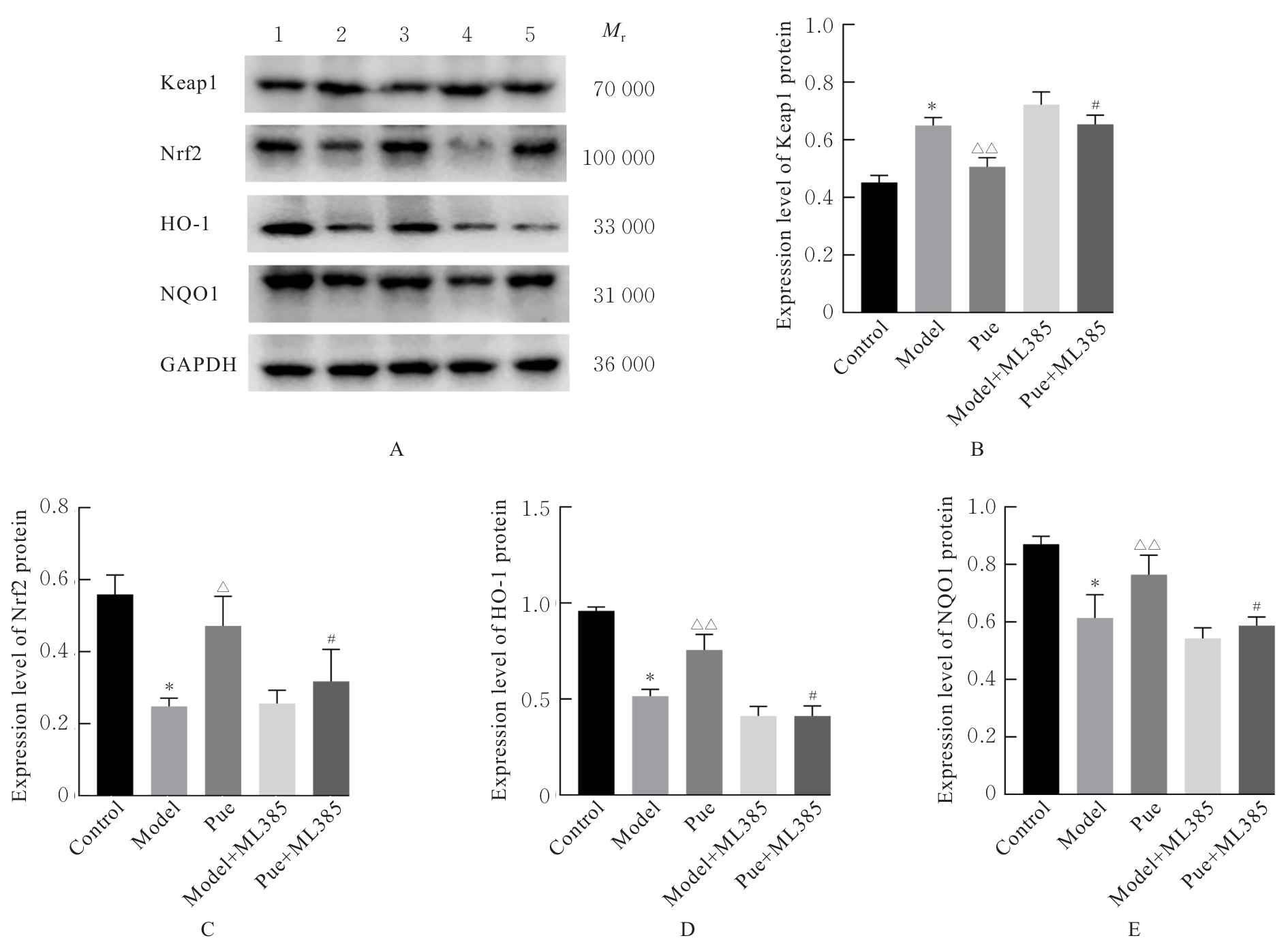
Levels of MDA and activities of SOD and GSH-Px in liver tissue of mice in various groups"
| Group | MDA [mB/(μmol·g-1)] | SOD [λB/(U·mg-1)] | GSH-Px [λB/(U·mg-1)] |
|---|---|---|---|
| Control | 1.39±0.13 | 139.82±7.06 | 127.09±4.67 |
| Model | 2.81±0.20* | 69.28±12.00* | 54.73±6.26* |
| Pue | 1.76±0.22△ | 110.59±9.10△ | 100.89±9.92△ |
| Model+ML385 | 3.01±0.25 | 58.45±6.39 | 43.17±7.38 |
| Pue+ML385 | 2.08±0.10# | 95.32±8.23# | 81.82±9.09## |
Tab.7
Levels of IL-6, IL-1β, and TNF-α in liver tissue of mice in various groups [n=6, x±s,ρB/(ng·L-1)]"
| Group | IL-6 | IL-1β | TNF-α |
|---|---|---|---|
| Control | 16.14±1.05 | 13.84±1.04 | 108.50±5.77 |
| Model | 28.80±1.44* | 31.07±1.56* | 202.23±9.51* |
| Pue | 22.65±3.20△ | 21.35±1.56△ | 143.33±7.78△ |
| Mod+ML385 | 31.15±2.06 | 34.52±1.72 | 214.49±5.76 |
| Pue+ML385 | 26.70±0.99# | 23.87±0.99# | 158.76±8.32# |
| [1] | AMORIM R, SOARES P, CHAVARRIA D, et al. Decreasing the burden of non-alcoholic fatty liver disease: From therapeutic targets to drug discovery opportunities [J]. Eur J Med Chem, 2024, 277(15): 116723. |
| [2] | WEI S, WANG L, EVANS P C, et al. NAFLD and NASH: etiology, targets and emerging therapies [J]. Drug Discov Today, 2024, 29(3): 103910. |
| [3] | HAN S K, BAIK S K, KIM M Y. Non-alcoholic fatty liver disease: Definition and subtypes [J]. Clin Mol Hepatol, 2023, 29(): S5-S16. |
| [4] | THIRUVENGADAM M, VENKIDASAMY B, SUBRAMANIAN U, et al. Bioactive compounds in oxidative stress-mediated diseases: targeting the NRF2/ARE signaling pathway and epigenetic regulation [J]. Antioxidants (Basel), 2021, 10(12):1895. |
| [5] | YANG K, YU X, GUO Z, et al. PIM1 alleviated liver oxidative stress and NAFLD by regulating the NRF2/HO-1/NQO1 pathway [J]. Life Sci, 2024, 349(14): 122714. |
| [6] | PENGNET S, SUMARITHUM P, PHONGNU N, et al. Naringin attenuates fructose-induced NAFLD progression in rats through reducing endogenous triglyceride synthesis and activating the Nrf2/HO-1 pathway [J]. Front Pharmacol, 2022, 13: 1049818. |
| [7] | WU Q, LI P, LI X, et al. Pueraria extract ameliorates alcoholic liver disease via the liver-gut-brain axis: focus on restoring the intestinal barrier and inhibiting alcohol metabolism [J]. J Agric Food Chem, 2024, 72(44): 24449-24462. |
| [8] | YANG M, XIA L, SONG J, et al. Puerarin ameliorates metabolic dysfunction-associated fatty liver disease by inhibiting ferroptosis and inflammation [J]. Lipids Health Dis, 2023, 22(1): 202-217. |
| [9] | YEN P T, HUANG S E, HSU J H, et al. Anti-Inflammatory and anti-oxidative effects of Puerarin in postmenopausal cardioprotection: roles of Akt and heme oxygenase-1[J]. Am J Chin Med, 2023, 51(1): 149-168. |
| [10] | ZHOU W, HE H, WEI Q, et al. Puerarin protects against acetaminophen-induced oxidative damage in liver through activation of the Keap1/Nrf2 signaling pathway [J]. Food Sci Nutr, 2023, 11(10): 6604-6615. |
| [11] | ZHOU J, ZHANG N, ALDHAHRANI A, et al. Puerarin ameliorates nonalcoholic fatty liver in rats by regulating hepatic lipid accumulation, oxidative stress, and inflammation [J]. Front Immunol, 2022, 13: 956688. |
| [12] | DINIZ T A, DE LIMA JUNIOR E A, TEIXEIRA A A, et al. Aerobic training improves NAFLD markers and insulin resistance through AMPK-PPAR-α signaling in obese mice [J]. Life Sci, 2021, 266: 118868. |
| [13] | LIU J, SUN J, YU J, et al. Gut microbiome determines therapeutic effects of OCA on NAFLD by modulating bile acid metabolism [J]. NPJ Biofilms Microbiomes, 2023, 9(1): 29-39. |
| [14] | GONG P, WANG J, WANG S, et al. Metabolomic analysis of the Puerarin hypoglycemic activity via AMPK-mTOR and PPARγ-NF-κB signaling pathways [J]. Phytomedicine, 2024, 130: 155546. |
| [15] | HEEREN J, SCHEJA L. Metabolic-associated fatty liver disease and lipoprotein metabolism [J]. Mol Metab, 2021, 50(8): 101238. |
| [16] | FLESSA C M, NASIRI-ANSARI N, KYROU I, et al. Genetic and diet-induced animal models for non-alcoholic fatty liver disease (NAFLD) research [J]. Int J Mol Sci, 2022, 23(24): 15791. |
| [17] | ZHENG Y, WANG S, WU J, et al. Mitochondrial metabolic dysfunction and non-alcoholic fatty liver disease: new insights from pathogenic mechanisms to clinically targeted therapy [J]. J Transl Med, 2023, 21(1): 510-530. |
| [18] | GUO X, YIN X, LIU Z, et al. Non-alcoholic fatty liver disease (NAFLD) pathogenesis and natural products for prevention and treatment [J]. Int J Mol Sci, 2022, 23(24): 15489. |
| [19] | ALLAMEH A, NIAYESH-MEHR R, ALIARAB A, et al. Oxidative stress in liver pathophysiology and disease [J]. Antioxidants (Basel), 2023, 12(9): 1653-1675. |
| [20] | ZHU L, LI B, CHEN D, et al. sSTEAP4 regulates cellular homeostasis and improves high-fat-diet-caused oxidative stress in hepatocytes [J]. Life Sci, 2022, 296(9): 120438. |
| [21] | KARKUCINSKA-WIECKOWSKA A, SIMOES I C M, KALINOWSKI P, et al. Mitochondria, oxidative stress and nonalcoholic fatty liver disease: A complex relationship[J]. Eur J Clin Invest, 2022, 52(3): e13622. |
| [22] | 李岫滟, 雷 娜, 宋虹霏, 等. 内质网应激在非酒精性脂肪性肝病中的作用及相关靶向治疗[J]. 临床肝胆病杂志, 2024, 40(11): 2300-2305. |
| [23] | FREDRICKSON G, BARROW F, DIETSCHE K, et al. Exercise of high intensity ameliorates hepatic inflammation and the progression of NASH[J]. Mol Metab, 2021, 53(11): 101270. |
| [24] | DUAN Y, PAN X, LUO J, et al. Association of inflammatory cytokines with non-alcoholic fatty liver disease[J]. Front Immunol, 2022, 13: 880298. |
| [25] | YANG C W, LIU H M, CHANG Z Y, et al. Puerarin modulates hepatic farnesoid X receptor and gut microbiota in high-fat diet-induced obese mice[J]. Int J Mol Sci, 2024, 25(10): 5274-5299. |
| [26] | LI L, YANG L, YANG L, et al. Network pharmacology: a bright guiding light on the way to explore the personalized precise medication of traditional Chinese medicine[J]. Chin Med, 2023, 18(1): 146-164. |
| [27] | 段明秀, 陈新利, 常伟宇, 等. 非酒精性脂肪性肝病多组学研究现状[J]. 临床肝胆病杂志, 2024, 40(6): 1240-1247. |
| [28] | ABDULAAL W H, OMAR U M, ZEYADI M, et al. Modulation of the crosstalk between Keap1/Nrf2/HO-1 and NF-κB signaling pathways by Tomatidine protects against inflammation/oxidative stress-driven fulminant hepatic failure in mice[J]. Int Immunopharmacol, 2024, 130: 111732. |
| [29] | DING C, WU Y, ZHAN C, et al. Research progress on the role and inhibitors of Keap1 signaling pathway in inflammation[J]. Int Immunopharmacol, 2024, 141(16): 112853. |
| [30] | LI B, NASSER M I, MASOOD M, et al. Efficiency of traditional Chinese medicine targeting the Nrf2/HO-1 signaling pathway[J]. Biomed Pharmacother, 2020, 126(6): 110074. |
| [31] | FANG Q L, QIAO X, YIN X Q, et al. Flavonoids from Scutellaria amoena C. H. Wright alleviate mitochondrial dysfunction and regulate oxidative stress via Keap1/Nrf2/HO-1 axis in rats with high-fat diet-induced nonalcoholic steatohepatitis[J]. Biomed Pharmacother, 2023, 158: 114160. |
| [32] | DONG H, ZHAO Y, TENG H, et al. Pueraria lobata antioxidant extract ameliorates non-alcoholic fatty liver by altering hepatic fat accumulation and oxidative stress[J]. J Ethnopharmacol, 2024, 333(19): 118468. |
| [1] | Ziyi TANG,Shiying YANG,Tianzhen YANG,Wenqiang LIU,Jiangxue ZHONG,Li YIN. Induction effect of pesticide pyraclostrobin on ferroptosis of spermatocytes GC-2 of mice [J]. Journal of Jilin University(Medicine Edition), 2026, 52(1): 18-25. |
| [2] | Huiyan ZHU,Min CHEN,Jinxian LI,Chunli LI. Effect of enriched environment on neurofunctional damage in rats with ischemic stroke via transcription factor EB-mediated autophagy [J]. Journal of Jilin University(Medicine Edition), 2026, 52(1): 116-124. |
| [3] | Tao SUN,Zhiyin DAI,Xuan LI,Chaopu ZHANG,Shu DING,Jianwei ZHAO. Predictive value of pan-immune-inflammation index for major adverse cardiovascular events within 1 year after PCI in elderly patients with coronary heart disease [J]. Journal of Jilin University(Medicine Edition), 2025, 51(6): 1655-1660. |
| [4] | Sifan FENG,Yunfeng LI,Jiaying WANG,Fubin MA,Yan WANG. Effects of heme-binding protein 1 gene knockdown on proliferation, migration, and inflammatory response of microglia BV2 and their mechanisms [J]. Journal of Jilin University(Medicine Edition), 2025, 51(6): 1532-1541. |
| [5] | Ying HU,Yong HUANG. Improvement effect of short-chain fatty acids on inflammation-induced autophagic damage in ovarian granulosa cells in polycystic ovary syndrome and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(5): 1340-1348. |
| [6] | Guang YANG,Zhifang ZHENG,Xinhua ZHANG. Research progress in protective effect of miRNA on neonatal hypoxic-ischemic brain injury [J]. Journal of Jilin University(Medicine Edition), 2025, 51(5): 1423-1428. |
| [7] | Yi ZHAO,Bing ZHOU,Huirui QIU,Xuan LI,Xiangli CUI. Improvement effect of lovastatin on hyperlipidemia-induced liver injury in rats and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(5): 1155-1164. |
| [8] | Nan LU,Mingxin DONG,Lei YU,Chengbiao SUN,Yan WANG,Na XU,Wensen LIU,Shumin GE. Transcriptome sequencing-based expression profiling of oxidative stress-related genes and circRNAs in ricin toxin-induced macrophage pyroptosis [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 1007-1018. |
| [9] | Lieqian SUN,Mengyu GU,Jie YANG,Kaiyi WANG,Gaoshuai GUO,Hongbo ZHANG,Siyi ZHANG,Tanglong WANG,Zhiwei YANG,Yanni HE,Chao YANG. Effect of bone marrow-derived mesenchymal stem cell transplantation on mitochondrial autophagy in rats with vascular dementia through ROS/Nrf2 signaling and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(3): 610-620. |
| [10] | Han LIN,Qiuyan YANG,Jieyue ZHONG,Bolun CHEN,Wangxia TONG. Improvement effect of cordycepin on ferroptosis in HepG2 cells induced by RSL3 and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(3): 576-589. |
| [11] | Yue WANG,Ning MA,Jiajun LU,Chengyao WANG,Linyu CHEN,Yuchen REN,Jingwu LI,Hong SUN. Protective effect of novel composite hydrogels on H₂O₂-induced oxidative stress injury in cardiomyocytes [J]. Journal of Jilin University(Medicine Edition), 2025, 51(2): 352-359. |
| [12] | Xiaoxia HU,Yalong LI,Dongliang YANG,Bazeren LA,Xinyue LIU. Effect of high glucose on polarization of Raw264.7 macrophages in vitro [J]. Journal of Jilin University(Medicine Edition), 2025, 51(2): 403-411. |
| [13] | Junjie JIANG,Hao WU,Kang HE,Zhiqiang SAN,Qing YANG,Hui LI,Na LI. Repair effect of ginseng polypeptide thermosensitive hydrogel on heat-induced skin injury in rats and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(2): 360-369. |
| [14] | Chaohe ZHANG,Xinwei ZHANG,Xiangfeng WANG. Protective effect of Pien-Tze-Huang on acetaminophen-induced liver injury and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(1): 105-114. |
| [15] | Xingqi SU,Lingmin ZHAO,Di MA,Jiulin YOU,Ying CHEN,Liangshu FENG,Jing WANG,Jiachun FENG,Chuan WANG. Analysis on correlation of cerebral infarct area with cytokines and immune status in patients with acute ischemic stroke [J]. Journal of Jilin University(Medicine Edition), 2025, 51(1): 124-132. |
|
||