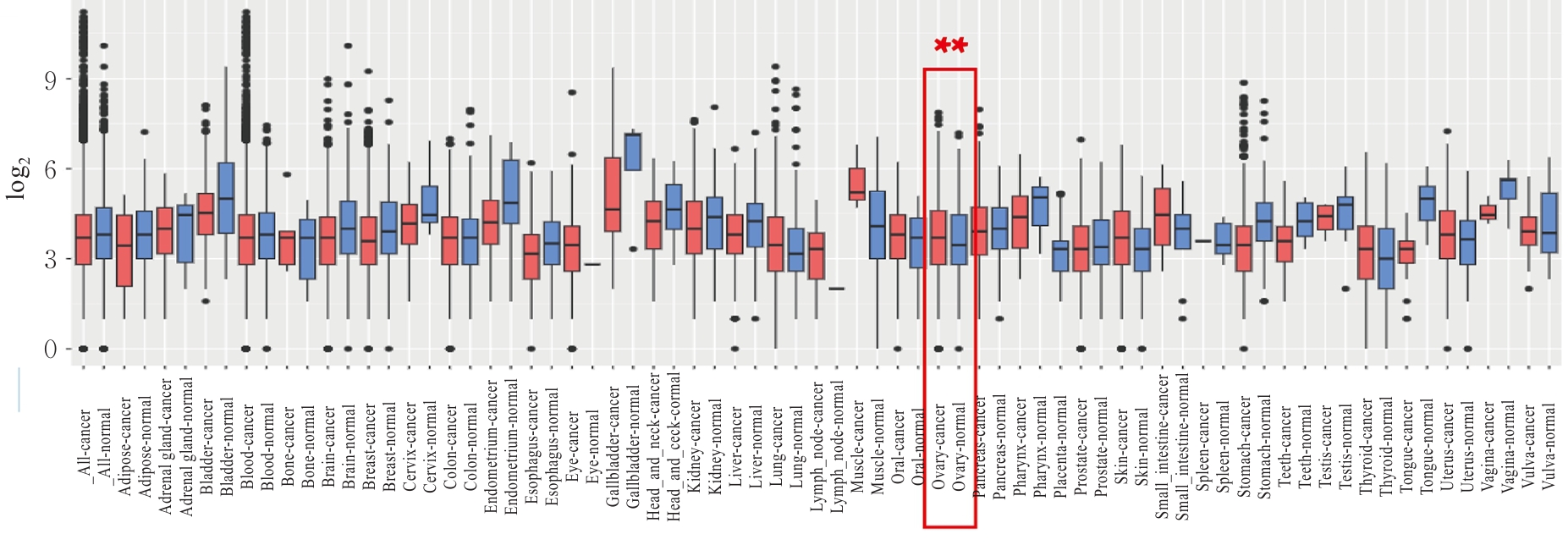
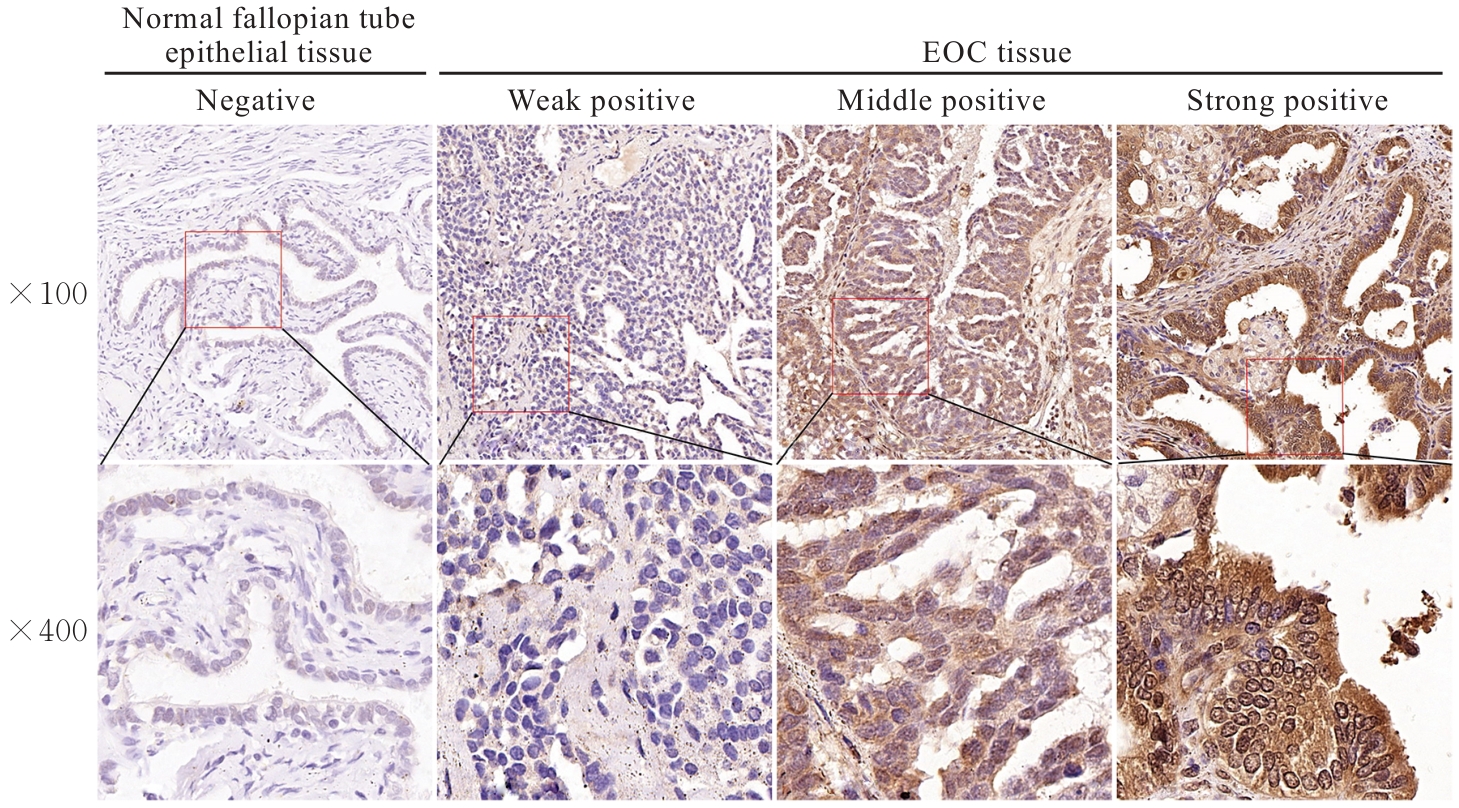
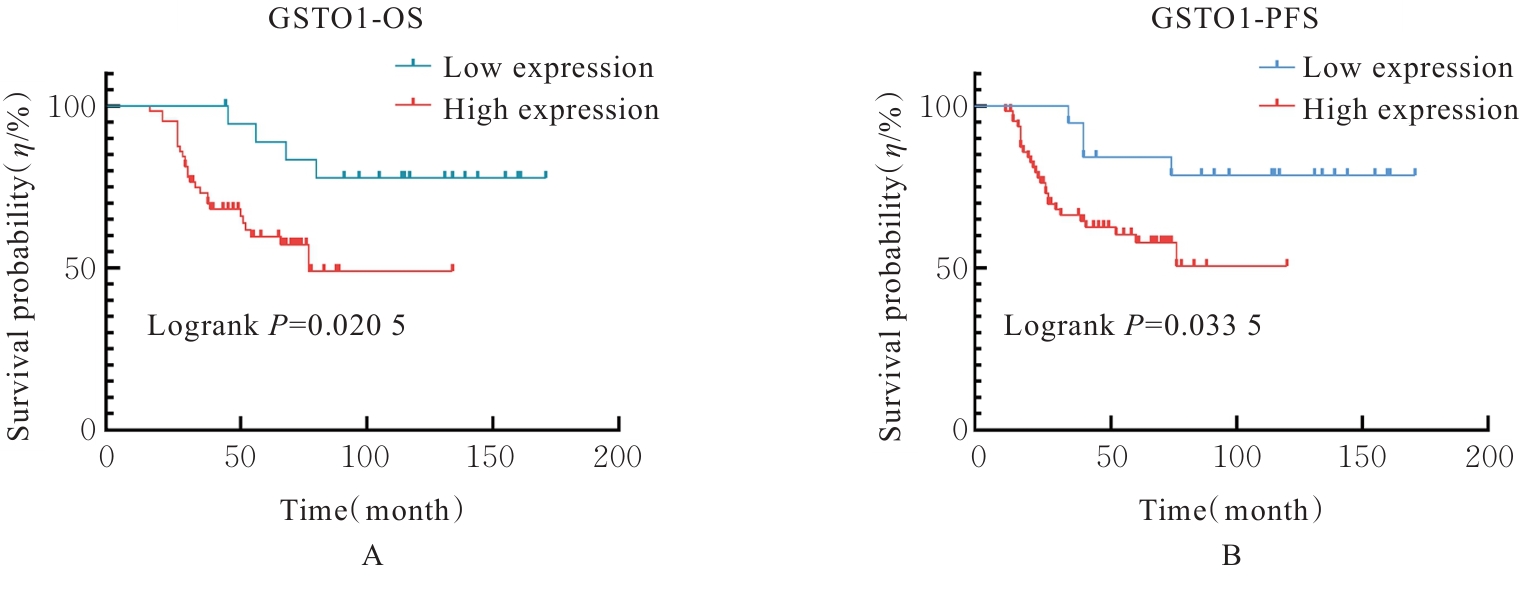
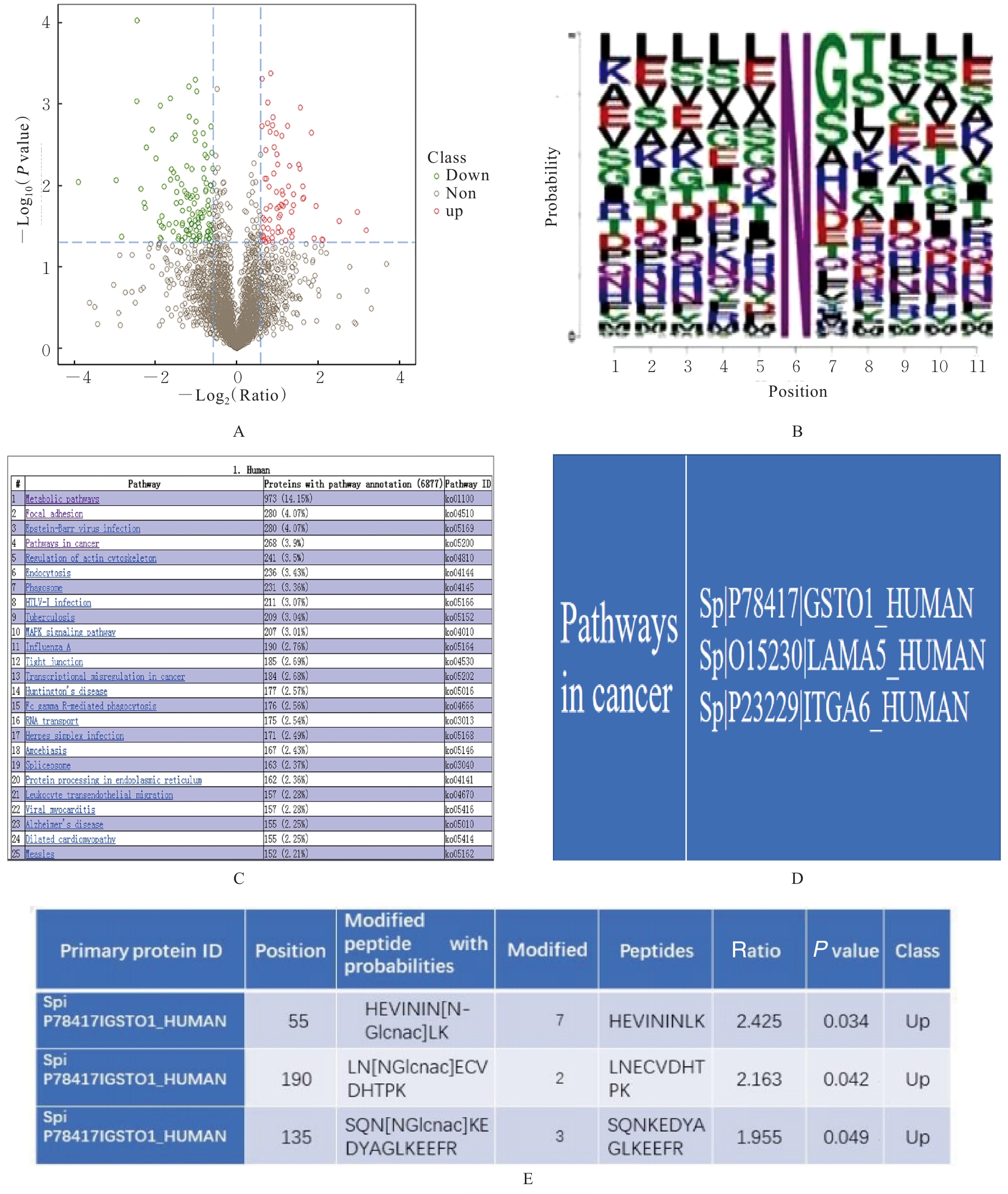
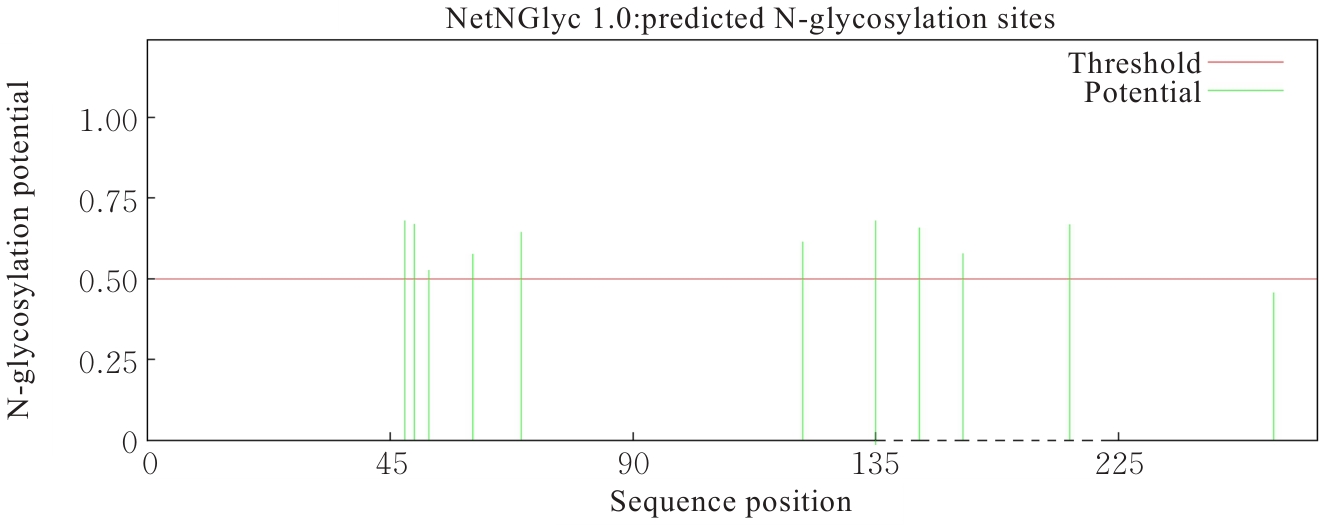
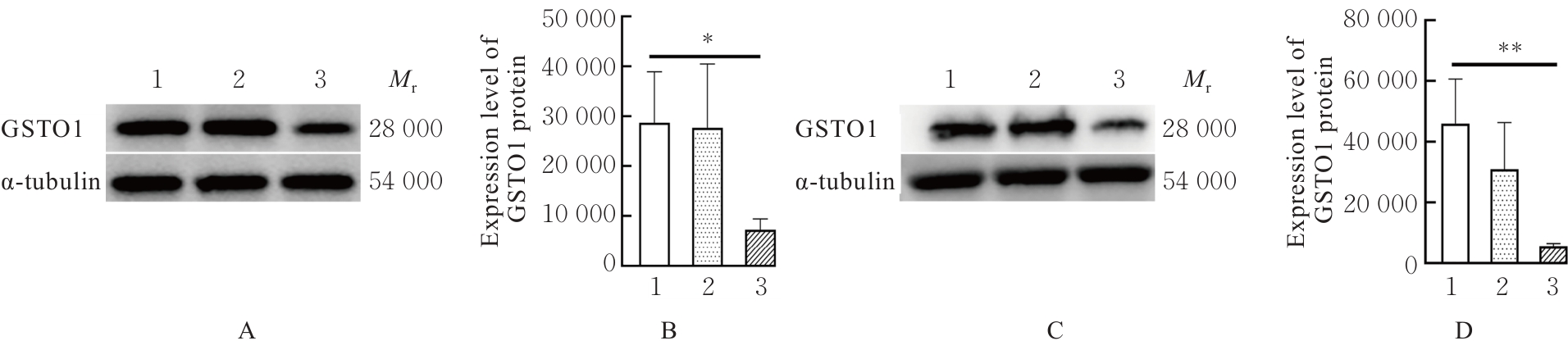
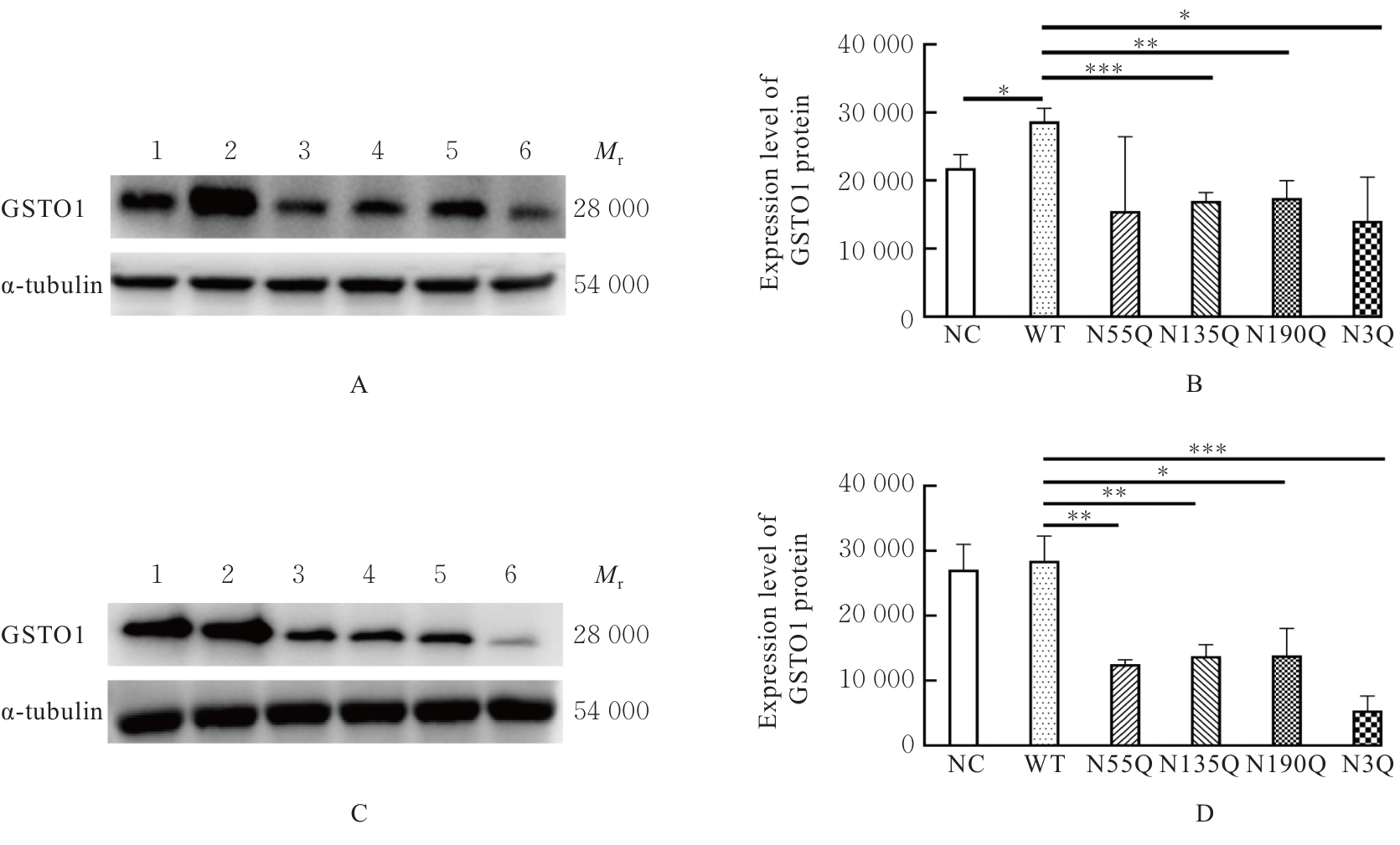
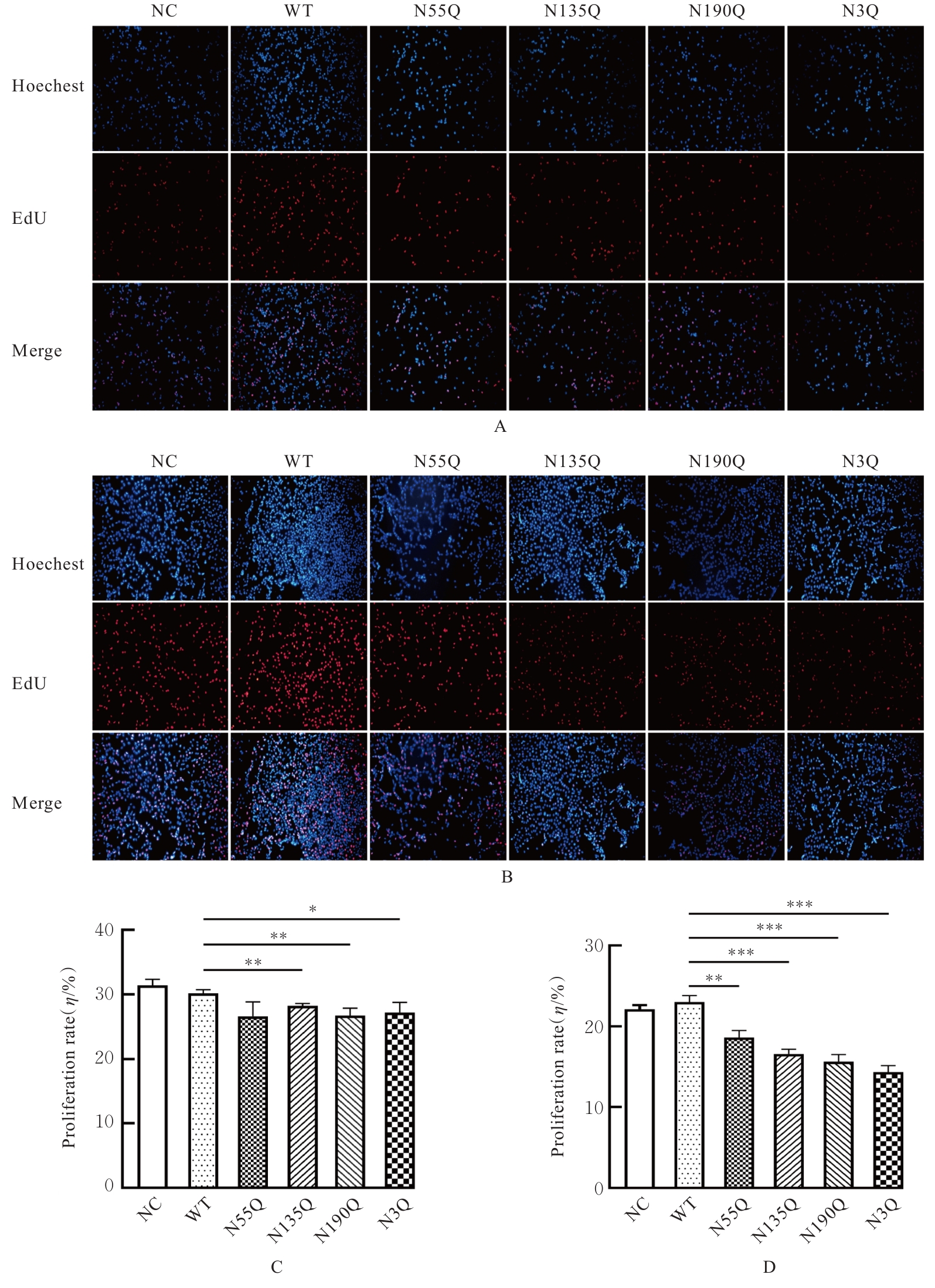
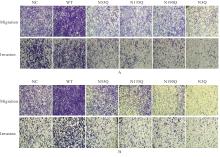
Journal of Jilin University(Medicine Edition) ›› 2026, Vol. 52 ›› Issue (2): 469-482.doi: 10.13481/j.1671-587X.20260219
• Research in clinical medicine • Previous Articles
Expression of GSTO1 in ovarian cancer tissue and effect of N-glycosylation site mutations on biological behaviors of epithelial ovarian cancer cells
Hong LI1,2,Panpan YU2,Zouyu ZHAO1,2,Chongfeng SUN1,2,Hui QIAO1,2,Ping YANG1,2,3( )
)
- 1.Department of Gynecology,First Affiliated Hospital,Shihezi University,Shihezi 832008,China
2.School of Medicine,Shihezi University,Shihezi 832008,China
3.Department of Gynecology,Xinjiang Production and Construction Corps Hospital,Urumqi 830001,China
-
Received:2025-04-25Accepted:2025-07-06Online:2026-03-28Published:2026-04-15 -
Contact:Ping YANG E-mail:pingy2018@163.com
CLC Number:
- R737.33
Cite this article
Hong LI,Panpan YU,Zouyu ZHAO,Chongfeng SUN,Hui QIAO,Ping YANG. Expression of GSTO1 in ovarian cancer tissue and effect of N-glycosylation site mutations on biological behaviors of epithelial ovarian cancer cells[J].Journal of Jilin University(Medicine Edition), 2026, 52(2): 469-482.
share this article
Tab.1
Baseline data of 88 patients with EOCn [(η/%)]"
| Characteristic | Number | |
|---|---|---|
| Age(year) | ||
| ≤52 | 48 (54.5) | |
| >52 | 40 (45.5) | |
| FIGO Stage(2014) | ||
| Ⅰ | 36 (41.0) | |
| Ⅱ | 12 (13.6) | |
| Ⅲ | 39 (44.3) | |
| Ⅳ | 1 (1.1) | |
| Pathological type | ||
| Plasmacytoid cancer | 65 (73.9) | |
| Mucinous carcinoma | 7 (8.0) | |
| Clear cell carcinoma | 6 (6.8) | |
| Endometrioid cancer | 9 (10.2) | |
| Mixed epithelial carcinoma | 1 (1.1) | |
| Grade | ||
| Low | 17 (19.3) | |
| Middle | 9 (10.2) | |
| High | 62 (70.5) | |
| LVSI | ||
| Yes | 24 (27.3) | |
| No | 64 (72.7) | |
| LNM | ||
| Yes | 29 (33.0) | |
| No | 59 (67.0) | |
| Size(cm) | ||
| ≤8 | 46 (52.3) | |
| >8 | 42 (47.7) | |
| CA125(U·mL-1) | ||
| ≤200 | 48 (54.5) | |
| >200 | 40 (45.5) |
Tab.2
Expressions of GSTO1 protein in ovarian tissue of EOC patients with different clinicopathological characteristics"
| Characteristic | n | GSTO1 | χ2 | P | |
|---|---|---|---|---|---|
| Low | High | ||||
| Age(year) | 0.244 | 0.621 | |||
| ≤52 | 48 | 13(27.1) | 35(72.9) | ||
| >52 | 40 | 9(22.5) | 31(77.5) | ||
| FIGO Stage(2014) | 6.111 | 0.013 | |||
| Ⅰ-Ⅱ | 48 | 17(35.4) | 31(64.6) | ||
| Ⅲ-Ⅳ | 40 | 5(12.5) | 35(87.5) | ||
| Pathological type | 0.491 | 0.484 | |||
| Plasmacytoid cancer | 65 | 15(23.1) | 50(76.9) | ||
| Adenocarcinoma | 23 | 7(30.4) | 16(69.6) | ||
| Grade | 0.024 | 0.876 | |||
| Low | 17 | 4 (23.5) | 13(76.5) | ||
| Other divisions | 71 | 18(25.4) | 53(74.6) | ||
| CA125(U·mL-1) | 0.244 | 0.621 | |||
| ≤200 | 48 | 13(27.1) | 35(72.9) | ||
| >200 | 40 | 9(22.5) | 31(77.5) | ||
| Size(cm) | 26.783 | <0.001* | |||
| ≤8 | 46 | 22(47.8) | 24(52.2) | ||
| >8 | 42 | 0(0.0) | 42(100.0) | ||
| LVSI | 4.889 | 0.027 | |||
| Yes | 24 | 10(41.7) | 14(58.3) | ||
| No | 64 | 12(18.8) | 52(81.2) | ||
| LNM | 0.017 | 0.896 | |||
| Yes | 29 | 7(24.1) | 22(75.9) | ||
| No | 59 | 15(25.4) | 44(74.6) | ||
Tab.3
Univariate and multivariate Cox regression analyses of influencing factors of OS in EOC patients"
| Characteristic | Univariate Cox analysis | Multivariate Cox analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | P | HR | 95%CI | P | |
| FIGO stage (Ⅲ-Ⅳ vs Ⅰ-Ⅱ) | 9.948 | 4.037-24.510 | 0.001 | 14.272 | 3.509-58.042 | 0.001 |
| Grade (High vs low) | 1.719 | 0.600-4.928 | 0.919 | |||
| Size (>8 cm vs ≤8 cm) | 4.698 | 2.030-10.873 | 0.001 | 0.516 | 0.113-2.345 | 0.392 |
| LVSI (Positive vs negative) | 1.999 | 0.974-4.103 | 0.059 | |||
| LNM (Positive vs negative) | 3.271 | 1.582-6.764 | 0.001 | 3.397 | 1.606-7.183 | 0.001 |
| GSTO1 (High vs low) | 3.376 | 1.138-10.017 | 0.028 | 4.881 | 1.024-23.270 | 0.029 |
Tab.4
Univariate and multivariate Cox regression analyses in influencing factors of PFS in EOC patients"
| Characteristics | Univariate Cox analysis | Multivariate Cox analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | P | HR | 95%CI | P | |
| FIGO stage (Ⅲ-Ⅳ vs Ⅰ-Ⅱ) | 8.109 | 3.765-17.462 | 0.001 | 8.468 | 2.765-25.933 | 0.001 |
| Grade (High vs low) | 1.427 | 0.597-3.408 | 0.424 | |||
| Size (>8 cm vs ≤8 cm) | 4.864 | 2.349-10.069 | 0.001 | 0.669 | 0.193-2.320 | 0.526 |
| LVSI (Positive vs negative) | 1.483 | 0.776-2.834 | 0.233 | |||
| LNM (Positive vs negative) | 2.306 | 1.220-4.358 | 0.010 | 2.583 | 1.335-14.588 | 0.005 |
| GSTO1 (High vs low) | 4.782 | 1.695-13.488 | 0.003 | 4.119 | 1.163-14.588 | 0.028 |
| [1] | LOKMAN N A, RICCIARDELLI C, STEPHENS A N, et al. Diagnostic value of plasma annexin A2 in early-stage high-grade serous ovarian cancer[J]. Diagnostics, 2021, 11(1): 69. |
| [2] | ZHANG R Q, SIU M K Y, NGAN H Y S, et al. Molecular biomarkers for the early detection of ovarian cancer[J]. Int J Mol Sci, 2022, 23(19): 12041. |
| [3] | FERNÁNDEZ L P, SÁNCHEZ-MARTÍNEZ R, VARGAS T, et al. The role of glycosyltransferase enzyme GCNT3 in colon and ovarian cancer prognosis and chemoresistance[J]. Sci Rep, 2018, 8(1): 8485. |
| [4] | AN H J, FROEHLICH J W, LEBRILLA C B. Determination of glycosylation sites and site-specific heterogeneity in glycoproteins[J]. Curr Opin Chem Biol, 2009, 13(4): 421-426. |
| [5] | ZHANG J, DIJKE PTEN, WUHRER M, et al. Role of glycosylation in TGF-β signaling and epithelial-to-mesenchymal transition in cancer[J]. Protein Cell, 2021, 12(2): 89-106. |
| [6] | 于盼盼, 杨 萍, 孙倩玉, 等. 宫颈癌组织GSTO1的表达与宫颈癌预后的相关性分析及其N-糖基化对宫颈癌恶性生物学行为的影响[J]. 安徽医科大学学报, 2023, 58(12): 2002-2010. |
| [7] | SIMIC P, CORIC V, PLJESA I, et al. The role of glutathione transferase omega-class variant alleles in individual susceptibility to ovarian cancer[J]. Int J Mol Sci, 2024, 25(9): 4986. |
| [8] | GUO J, CAI J, ZHANG Y X, et al. Establishment of two ovarian cancer orthotopic xenograft mouse models for in vivo imaging: a comparative study[J]. Int J Oncol, 2017, 51(4): 1199-1208. |
| [9] | 孙崇凤, 杨萍, 侯纪帅, 等. STT3A和STT3B在上皮性卵巢癌中的表达及与预后的关系[J]. 实用医学杂志, 2023, 39(16): 2062-2070. |
| [10] | YANG C, XIA B R, ZHANG Z C, et al. Immunotherapy for ovarian cancer: adjuvant, combination, and neoadjuvant[J]. Front Immunol, 2020, 11: 577869. |
| [11] | XU Y B, BANKHEAD A 3rd, TIAN X L, et al. Deletion of glutathione S-transferase omega 1 activates type Ⅰ interferon genes and downregulates tissue factor[J]. Cancer Res, 2020, 80(17): 3692-3705. |
| [12] | PETROVIC M, SIMIC T, DJUKIC T, et al. The polymorphisms in GSTO genes (GSTO1 rs4925, GSTO2 rs156697, and GSTO2 rs2297235) affect the risk for testicular germ cell tumor development: a pilot study[J]. Life, 2023, 13(6): 1269. |
| [13] | WANG L K, YUE H L, PENG X J, et al. GSTO1 regards as a meritorious regulator in cutaneous malignant melanoma cells[J]. Mol Cell Probes, 2019, 48: 101449. |
| [14] | MA H, CHEN X L, MO S W, et al. Targeting N-glycosylation of 4F2hc mediated by glycosyltransferase B3GNT3 sensitizes ferroptosis of pancreatic ductal adenocarcinoma[J]. Cell Death Differ, 2023, 30(8): 1988-2004. |
| [15] | HE L N, GUO Z J, WANG W J, et al. FUT2 inhibits the EMT and metastasis of colorectal cancer by increasing LRP1 fucosylation[J]. Cell Commun Signal, 2023, 21(1): 63. |
| [16] | 井玉莹, 杨凯歌, 程仪婷, 等. EZH2通过EMT促进食管鳞状细胞癌的恶性生物学行为[J]. 中南大学学报(医学版), 2025, 50(2): 155-166. |
| [17] | PARK M, KIM D, KO S, et al. Breast cancer metastasis: mechanisms and therapeutic implications[J]. Int J Mol Sci, 2022, 23(12): 6806. |
| [18] | BABAEI G, AZIZ S G, JAGHI N Z Z. EMT, cancer stem cells and autophagy; The three main axes of metastasis[J]. Biomed Pharmacother, 2021, 133: 110909. |
| [19] | 张 哲, 罗怡欣, 陈 卓, 等. 中性粒细胞源性外泌体在乳腺癌细胞侵袭、 转移中的作用[J]. 郑州大学学报(医学版), 2025, 60(4): 484-487. |
| [20] | 王姜婷, 孙 恺, 高 谋, 等. SIRT7调控上皮-间质转化对胶质瘤细胞增殖和迁移的作用[J]. 解放军医学杂志, 2025, 50(1): 57-68. |
| [21] | 李瑛花, 杨 娟. LncRNA RP11-499E18.1对卵巢癌细胞恶性生物学行为的影响[J]. 中南大学学报(医学版), 2025, 50(1): 1-10. |
| [22] | 王发辉, 邓青春, 林佳佳, 等. GATA3介导miR-21/PTEN轴对子宫内膜癌细胞增殖、侵袭的影响[J]. 实用医学杂志, 2024, 40(15): 2069-2074. |
| [1] | Yao ZHENG,Mingxia FU,Weichen WANG,Weiwei CHEN,Yuchen HAN,Yu BAI,Jiajia AN. Effect of sodium selenite on biological behaviors of breast cancer doxorubicin-resistant MCF-7/ADR cells [J]. Journal of Jilin University(Medicine Edition), 2026, 52(2): 410-417. |
| [2] | Li ZHAI,Meng CHEN,Jianbo LUO,Aili ZHANG,Liangxiao WANG,Ying WEI,Xi ZHANG. Targeting relationship between miR-214-3p and EZH2 and its effects on proliferation, invasion, and apoptosis of ovarian cancer SKOV3 cells [J]. Journal of Jilin University(Medicine Edition), 2026, 52(2): 460-468. |
| [3] | Yunshan DING,Haitao DAI,Min CHEN,Xiaohui HAO,Xiao ZHOU,Nan WU. Effect of silencing TRPV2 gene and cannabidiol on biological behaviors of oral squamous cell carcinoma CAL-27 cells [J]. Journal of Jilin University(Medicine Edition), 2026, 52(1): 143-151. |
| [4] | Yan ZHAO,Huawei WU. Inhibitory effect of inhibition of miR-17-5p on proliferation of spinal cord astrocytes of rats induced by scratch injury through targeting Mfn2 expression [J]. Journal of Jilin University(Medicine Edition), 2026, 52(1): 81-92. |
| [5] | Xiaohan YAO,Mingchen YAO,Zhiqing WANG,Heyang LI,Yan YAN,Ningjing LEI. Effect of silencing GPR139 gene on proliferation, apoptosis and autophagy of breast cancer cells and its mechanism [J]. Journal of Jilin University(Medicine Edition), 2026, 52(1): 1-9. |
| [6] | Sifan FENG,Yunfeng LI,Jiaying WANG,Fubin MA,Yan WANG. Effects of heme-binding protein 1 gene knockdown on proliferation, migration, and inflammatory response of microglia BV2 and their mechanisms [J]. Journal of Jilin University(Medicine Edition), 2025, 51(6): 1532-1541. |
| [7] | Lulu FU,Yinggang ZOU,Xiaoyu ZHENG,Xueying ZHANG,Jingshun ZHANG,Min WANG,Qiang ZHANG,Lianwen ZHENG. Expression of placenta expressed transcription factor 1 in ovarian tissue of polycystic ovary syndrome rats and its effect on proliferation of rat ovarian granulosa cells [J]. Journal of Jilin University(Medicine Edition), 2025, 51(5): 1177-1184. |
| [8] | Pingsheng ZHU,Sitang GE,Lugen ZUO,Deli CHEN,Yangyang ZHANG. Effect of miR-325-3p targeting PRELID1 gene in regulation of EMT pathway on invasion and migration of colon cancer cells and their mechanisms [J]. Journal of Jilin University(Medicine Edition), 2025, 51(5): 1185-1193. |
| [9] | Wenxuan LI,Minru ZONG. Research progress in role of migration of Schwann cells in repairment of peripheral nerve injury [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 1137-1144. |
| [10] | Zhongjun SHEN,Yao ZHAO,Mingbo JIA,Liyan ZHAO. Research progress in effects of hypoxia-inducible factors on cell migration and invasion during epithelial-mesenchymal transition in glioma cells [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 1145-1154. |
| [11] | Yixuan GAO,Peng WANG,Silong ZHANG,Ruijuan GAO,Yingfang MA,Keke ZHANG,Dan FENG,Zongqi HUANG,Ketao MA,Li LI,Junqiang SI. Inhibitory effect of safranal on proliferation, migration and phenotypic transformation of vascular smooth muscle cells of rats induced by high glucose in vitro [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 948-957. |
| [12] | Xiaoshuang HE,Lina XU,Mei CUI,Yu ZHAO,Bei WANG,Zheng HUANG,Yuchao WANG,Wenyan XIN,Chao WU. Effects of lncRNA DUXAP8 in lung cancer A549 cells-derived exosomes on lung cancer cell growth and its mechnism [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 958-967. |
| [13] | Hongli LI,Mengyao WANG,Yangyang LIU,Hui ZHANG,Li LI. Effect of KHSRP on biological behavior of colorectal cancer cells through activation of JAK/STAT signaling pathway [J]. Journal of Jilin University(Medicine Edition), 2025, 51(4): 996-1006. |
| [14] | Xingxiang WANG,Ying ZHAO,Qiaotong REN,Hefei WANG,Gang PU,Chun LI. Promotive effect of M2 macrophages on epithelial-mesenchymal transition and cisplatin resistance in non-small cell lung cancer A549 cells by regulating NF-κB signaling pathway [J]. Journal of Jilin University(Medicine Edition), 2025, 51(3): 642-652. |
| [15] | Donghui LIU,Yunzhe CI,Chunyan WANG,Wenyi MA. Effect of miR-199a-5p on expression of Caveolin-1, cell migration and apoptosis in glioma U251 cells [J]. Journal of Jilin University(Medicine Edition), 2025, 51(3): 663-671. |
|
||